Amniotic membrane: An innovative treatment of refractory vascular ulcers?
Mireille DROUET**
Marie-Claire ROUSANNE***
Philippe LACROIX*
Vasculaire et Angiologie, CHU Dupuytren,
Limoges, France
** Service d’Immunologie, CHU Dupuytren,
Limoges, France
*** Etablissement français du sang Aquitaine-
Limousin
ABSTRACT
Mixed arterial and venous ulcers often heal poorly if there is no revascularization, as observed in the elderly, diabetics, and chronic renal failure patients who often have diffuse, distal, and calcified vascular lesions. Treatment therefore is based on optimized local wound care, pain control, and management of the risk of infection inherent in these types of patients with chronic wounds.
Current avenues of research in treatment are based on the development of angiogenesis and the use of growth factors to promote healing. Placental tissue is known to contain a large quantity of growth factors. Use of the latter was indirectly reported by Davis in 1910 and Sabella in 1913, who described the use of fresh amniotic membrane (AM) in the management of chronic wounds occurring in the aftermath of burns.1,2 Since then, many studies have reported the use of placental tissue or AM in the management of chronic wounds. Currently, AM is used after freezing in the management of corneal defects.
The aim of this article is to provide an updated literature review of this technique and its therapeutic prospects in the management of refractory vascular ulcers.
INTRODUCTION
Vascular ulcers are the leading cause of chronic skin changes in the industrialized world. Venous disease is the cause of 60% to 80% of such ulcers; 10% are purely arterial and 20% to 30% are mixed arterial/venous ulcers.3
Many studies have reported epidemiological data and the therapeutic management of venous ulcers. Few studies of arterial or mixed ulcers are currently available, yet, with the aging of the population, such ulcers are increasingly common. Their prevalence is at least 1% in subjects over age 65 years4,5 and approximately 1.41 per thousand in the general population.6-8
Revascularization is not always feasible because of the diffuse, distal features of vascular lesions in the elderly and frequent comorbidity factors, which can contraindicate surgical management. In spite of the development of many types of wound dressings, healing of such ulcers is often difficult to achieve and the diagnosis of peripheral arterial disease is not always established. Current avenues of research are directed at cell therapy and the use of growth factors to promote angiogenesis and the healing process. Some studies have reported that autologous stem cells and growth factors stimulate angiogenesis, suggesting a possible approach to healing in the event of chronic skin changes.
The placenta naturally contains high amounts of growth factors and was used in healing ointment formulations up to the 1980s. It continues to be used successfully for healing purposes in some countries such as India. For reasons related to bacterial and viral safety, it is no longer used. Davis in 1910 and Sabella in 1913 reported the value of placenta in healing.1,2 In 1980, Bennett, Matthews, and Faulk reported the use of fresh AM in the management of leg ulcers as a preparation for skin grafting.9,10 Considering the risk of bacterial and, in particular, viral infection, these techniques have not been developed.
However, AM can be frozen and serologically tested, thus enabling its possible use in therapeutic management. Currently, AM is used successfully in ophthalmology,11,12 and its use could perhaps be extended to the treatment of chronic skin changes, in particular arterial or mixed vascular ulcers. Here we provide an update on this therapy and its indications, mechanisms of action, and potential advantages in healing of ulcers, by means of a literature review and the authors’ own preliminary study.
MATERIAL
AM is histologically analogous to cutaneous tissue, and according to some authors is a fetal derivative of ectoderm.13 It consists of a cuboidal cell epithelium and a stroma resting on a basement membrane. It has no nerves, blood vessels, or lymphatics. AM expresses few antigens, which accounts for its good tolerability and the absence of rejection reactions.14
Becchetti et al reported AM’s high glycosaminoglycan content and that it contains hyaluronic acid, chondroitin sulfate, dermatane sulfate, and heparan sulfates.15 Their role in healing is now established, in particular that of hyaluronic acid, which is present in large quantities in fetal tissue and can produce complete healing.
Several authors have reported that AM may have bacteriostatic or bactericidal activity. Matthews et al reported a decrease in bacterial contamination of wounds treated with AM.16 Some authors consider that the bacteriostatic properties may simply be linked to the protective action of the membrane itself.14,17 Others believe that proteins such as allantoin, immunoglobulins, and lysozyme may be involved.18
Faulk et al have reported the benefits of AM in wound management.10 Histological analysis of leg ulcers before and 5 days after treatment with AM revealed an increase in granulation tissue and in connective tissue, formation of basement membrane, and vascular development. Increased expression of factor VIII synthesized by endothelial cells was also observed.10 Somerville has reported the formation of capillary neovessels following use of AM after storage at 4°C.18 AM expresses many neurotrophic and angiogenic factors: endothelins 2 and 3, vascular endothelial growth factor (VEGF), VEGF-B, Tie-2 angiopoietin receptor, ephrin-A2, ephrin receptors A2, B1, B3, B4, B5, neuropilin-2, nerve growth factor (NGF) receptor, and semaphorin-F19 as well as erythropoietin and its receptor.20,21
Histology studies of the cornea treated with AM indicate that the AM is resorbed, except for its basement membrane from which the cornea re-epithelializes, without a stromal, vascular, or inflammatory reaction.22 In summary, literature reports indicate that AM promotes healing by facilitating the formation of granulation tissue, an antibacterial action, and an antiinflammatory and analgesic action, without any evidence of rejection because the immunogenicity of AM is low.23
Currently, human AM is prepared from placenta obtained after scheduled delivery by cesarean section following a normal pregnancy. One placenta can provide 40 to 50 AM tissue fragments 5 cm in diameter. Currently, five centers in France process AM, each using its own techniques.
In our center, preparation is performed in a classified (class D) room with a microbiological safety workstation (class A), within 2 hours of a cesarean section delivery. The placenta is left in contact for 2 hours with an antibiotic solution in its collection container and is then rinsed with physiological saline and the AM is separated from the lamina propria and dissected. Nitrocellulose disks are placed on the membrane and the operator cuts around the disk. Thus, AM tissue fragments about 5 cm in diameter are obtained.
The membrane and the disk are rolled up and the entire structure is immersed in a sterile immediate packaging tube. This tube contains a cryoprotective agent (RPMI and glycerol). The primary tube is inserted into a secondary tube which also is sterile. Each tube is packaged in a hermetically sealed plastic bag. The product is stored in the freezer at a temperature of -80°C for up to 2 years (Figures 1 and 2). Three AM samples are collected for bacteriological examination. The placenta rinse fluid (8 to 10 mL) is used to inoculate 2 vials of aerobic and anaerobic organisms for bacteriological tests. The placenta is prepared for a pathology examination.
On the day of the cesarean section, test tubes containing blood from the mother are collected for the following serology tests: HIV 1 and 2: 2 tests, Ag p24, HCV, HTLV, syphilis: VDRL-TPHA, HBV: HBs antigen-HBc antibody. Final validation of the AM is performed after a repeat serology test by summoning the woman donor after 120 days.
AM is obtained on a doctor’s prescription, and kept on dry ice for transport. AM can be stored on dry ice for 24 hours and conserved 2 hours before utilization after thawing.

Figure 1: Processing of amniotic membrane.
Figure 2: Amniotic membrane in its preserving fluid
RESULTS
The use of AM in wound healing has been reported in the literature for almost a century,1,2 and its safety and excellent tolerability appear established: to date, no study has reported any episode of intolerance or allergy, probably because of its low immunogenicity.14,22
The usefulness of AM in wound healing is based on several mechanisms of action. First, AM acts as a dressing and isolates the wound, thus limiting the loss of protein and fluid and providing a medium that is conducive to healing. AM’s bacteriostatic action may result from this protective property and also from the expression of proteins with antibacterial activity, such as allantoin, lysozyme, and immunoglobulins.14,16,17,19 AM promotes healing and possibly angiogenesis also.10,18,21,23
AM has been studied in clinical practice with different types of wounds, such as burns, venous ulcers, radiation lesions, and arterial ulcers. Few clinical studies have been conducted on leg ulcers. The literature contains reports mainly on results obtained in ophthalmology.
Egan et al reported a study on 18 patients with venous ulcers of at least 8 years’ duration and treated with AM in preparation for skin grafting. Treatment with fresh AM shortened the time before skin grafting from 24.9 to 10.7 days by improvement of granulation tissue and vascularization of ulcers. An analgesic action was also noted. On the other hand, the rate of recurrence and delay to recurrence of an ulcer did not improve after prior treatment with AM.24
Ward et al demonstrated the optimization of skin grafting for venous ulcers after wound preparation with AM. 25 But the results appeared to vary depending on the methods of use and storage of AM. Thus, the skin graft failure rate was 6.3% when lyophilized AM was used, 11% with fresh amnion, and 25% when used after refrigeration. Mean healing time after skin grafting was also improved by preparation with AM, but depended on the storage method. Thus, mean time to healing was 22 days with lyophilized AM, 25 days with refrigerated AM, and 27.9 days with fresh amniotic membrane.25
Pretreatment of venous ulcers with AM prior to skin grafting did not seem to have an impact on the subsequent outcome of these ulcers after they healed, since the recurrence rate was estimated at 50% at one year.26
Hernandez et al reported efficacy in terms of healing in a heterogeneous group of patients with chronic wounds including burns, eschars, and venous ulcers. Fresh AM appeared beneficial, especially for eschars and burns.27 Encouraging results were noted in another study in 15 patients with arterial ulcers: one patient underwent amputation and 14 patients improved with local wound care performed with AM, with complete healing in 8.9 Analgesic and bacteriostatic actions were also observed after treatment of postthrombotic venous ulcers with AM used after storage at 4°C.28
In a preliminary study, we offered local wound care with frozen AM to 8 patients (7 men, 1 woman; mean age 69.5+/-9.6 years) with refractory vascular ulcers. Three patients had previously undergone arterial revascularization as well as treatment with wound dressings containing autologous keratinocytes (Epibase®) without improvement in their ulcers. Of the 8 patients, 4 had arterial lesions and 4 mixed ulcers. Mean ankle brachial pressure index was 0.62 +/- 0.24 (0.3 to 1). Transcutaneous oxygen pressure at baseline and after stimulation was 34 +/- 17 (1 to 64) mm Hg and 47 +/- 16 (26 to 74) mm Hg, respectively. The lesions were of at least 6 months’ duration. AM was placed in contact with the ulcer and held in place with secondary dressings; the former was changed once a week and the latter three times (Figures 3 to 8).
Figure 3: Preparation of the amniotic membrane and the wound
dressing interface before placement of a wound dressing.
Figure 4: Amniotic membrane
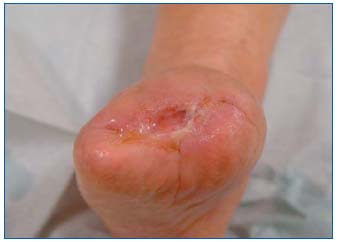
Figure 5: Application of the amniotic membrane to the wound.
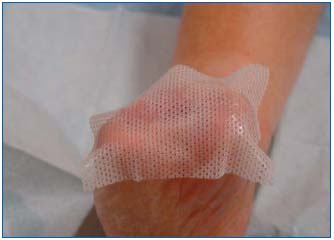
Figure 6: Application of a supporting interface
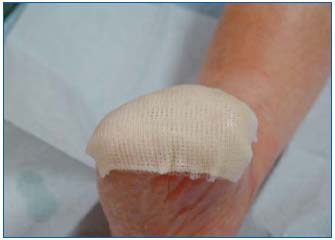
Figure 7: Application of an oily interface
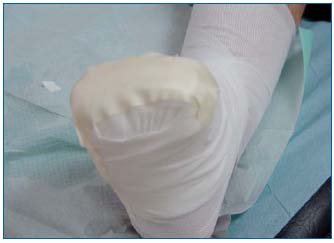
Figure 8: Securing of amniotic membrane and the interface with
a bandage
Complete wound healing was obtained in 2 patients after 19 and 26 weeks (Figures 9 and 10). Reduction in wound size of at least 50% was obtained in another 3 patients after 26, 31, and 32 weeks. One patient improved, but with no major reduction in wound area after 14 weeks. Two patients did not improve with this treatment.
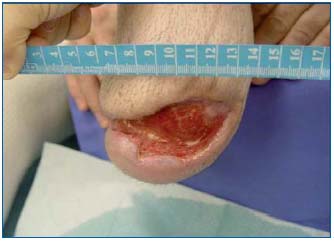
Figure 9: Delayed healing in a patient with peripheral arterial
disease: stump wound 6 months after leg amputation
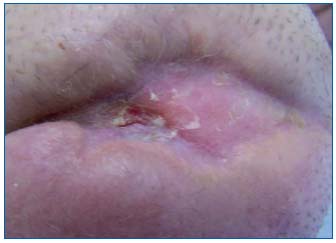
Figure 10: Outcome after 18 weeks of treatment with AM of the
amputation stump
No patient presented with intolerance or an adverse event after application of the AM. No patient presented with an infectious complication or an inflammatory syndrome during the duration of this treatment. No patient required antibiotic therapy. No patient underwent an amputation.
One of the patients who had complete healing presented with an ulcer recurrence after the end of treatment.
An analgesic effect was observed in 7 patients. The mean visual analogue score decreased from 4 at D0 to 1 at D90 (P=0.018).
Evaluation of the SF-36 quality of life questionnaire demonstrated a significant improvement in the perception of general health (P=0.01), a benefit for “relations with others”, which was at the limit of significance, as well as a benefit in “physical pain” (P=0.13),29 which was also at the limit of significance.
DISCUSSION
Literature data seem to establish the tolerability and safety of AM in ophthalmology, where it is commonly used to treat thermal or chemical burns, eye injury in Stevens-Johnson syndrome, and pemphigoid, and is also used in pterygium surgery and limbic transplantation,30 but also in the management of vascular leg ulcers. This excellent tolerability is probably related to its low immunogenicity, which is due to its low expression or nonexpression of the major histocompatibility complex antigens.20
The usefulness of AM lies in its analgesic and bacteriostatic actions, as reported in several studies, and this was true regardless of the method of preparation, ie, fresh, refrigerated, or frozen AM.
AM also has anti-inflammatory properties which can prove useful in the management of chronic wounds where there is often an imbalance between proteases and antiproteases. In fact, AM expresses an interleukin- 1 receptor antagonist, inhibitors of metalloproteinases 1,2, 3 and 4, and interleukin 10.20 Recent studies suggest that amniotic epithelial cells may have plasticity allowing differentiation into ecto-, meso-, or endodermal cells. In fact, they express common markers found on pluripotent stem cells, and can differentiate into hepatocytes, cardiac cells, and neurons.20,30,31 Their use for therapeutic purposes to regenerate tissues does not raise any ethical issues as with embryonic stem cells, since placental tissue is taken to be a byproduct of surgery.
The pro-angiogenic effect of AM is currently debated and it is unlikely that any such effect is involved in wound healing. Some ophthalmology studies have even suggested an anti-angiogenic action.32
AM is subject to traceability requirements and serological testing to ensure its safety for use in clinical practice. Freezing allows optimized storage and improves availability of AM. This method of storage preserves cell viability as well as the properties of growth factors and cytokines.31 New methods for storage of AM are now under evaluation. These involve infrared and microwave drying processes and sterilization with gamma radiation, which has the advantage of allowing extended storage at room temperature. Clinical studies are necessary to evaluate the efficiency of AM treated this way.20
AM promotes healing of injuries to the cornea, which is fragile tissue because of its fine blood vessels. Some studies suggest that AM promotes healing of chronic wounds, in particular venous or arterial vascular ulcers. However, such studies are often prospective, patients were not randomized, and patient populations are poorly defined and heterogeneous. Furthermore, most studies are old and report the use of fresh AM, AM refrigerated at 4°C or -25°C, or AM lyophilized and sterilized with gamma radiation.17,19,25 Currently, AM is used after freezing at -80°C. In this form, results are only available in ophthalmology. Few studies have examined the clinical utility of AM in the management of chronic wounds, in particular vascular ulcers. No study has reported poor tolerability or an increased risk of infection associated with AM.
Prospective studies on groups of homogeneous patients are necessary to establish the potential benefit of AM in the healing of vascular ulcers. Dissemination of this method will most probably be limited by the cost and availability of AM.
CONCLUSION
Management of chronic wounds using AM has a long history, but was abandoned because of the risk of bacterial and viral infection. AM can now be made safe for use by current methods of processing by freezing, and bacteriological and serological testing. AM is well tolerated when used for wound coverage, limits the risk of infection, and also has analgesic and antiinflammatory actions. AM has proved valuable in ophthalmology in the healing of corneal wounds, but its healing properties in vascular ulcers and other chronic wounds remain to be evaluated by prospective studies in homogeneous groups of patients. Extension of the therapeutic use of AM will, however, be limited by cost and availability.

REFERENCES
2. Sabella N. Use of the fetal membranes in skin grafting. Medical Record. 1913;15:478-480.
3. Cornwall JV, Doré CJ, Lewis JD. Leg ulcers: epidemiology and aetiology. Br J Surg. 1986;73:693-696.
4. Andersson E, Hansson C, Swanbeck G. Leg and foot ulcer prevalence and investigation of the peripheral arterial and venous circulation in a randomised elderly population. An epidemiological survey and clinical investigation. Acta Derm Venereol. 1993;73:57-61.
5. O’Brien JF, Grace PA, Perry IJ, Burke PE. Prevalence and aetiology of leg ulcers in Ireland. Ir J Med Sci. 2000;169:110-112.
6. Moffatt CJ, Franks PJ, Doherty DC, Martin R, Blewett R, Ross F. Prevalence of leg ulceration in a London population. QJM. 2004;97:431-437.
7. Pina E, Furtado K, Franks PJ, Moffatt CJ. Leg ulcer in Portugal: an underestimated health care problem. Rev Port Cir Cardiotorac Vasc. 2004;11:217-221.
8. Baker SR, Stacey MC, Singh G, Hoskin SE, Thompson PJ. Aetiology of chronic leg ulcers. Eur J Vasc Surg. 1992;6:245- 251.
9. Bennett JP, Matthews R, Faulk WP. Treatment of chronic ulceration of the legs with human amnion. Lancet. 1980;31:1153-1155.
10. Faulk WP, Matthews R, Stevens PJ, Bennett JP, Burgos H, His BL. Human amnion as an adjunct in wound healing. Lancet. 1980;31:1156-1157.
11. Muraine M, Descargues G, Franck O, et al. Amniotic membrane graft in ocular surface disease. Prospective study with 31 cases. J Fr Ophtalmol. 2001;24:798-812.
12. Von Versen-Hoynck F, Hesselbarth U, Moller DE. Application of sterilised human amnion for reconstruction of the ocular surface. Cell Tissue Bank. 2004;5:57-65.
13. Pigeon J. Treatment of second degree burns with amniotic membranes. Can Med Assoc J. 1960;83:844-845.
14. Singh R, Chouhan US, Purohit S, et al. Radiation processed amniotic membranes in the treatment of nonhealing ulcers of different etiologies. Cell Tissue Bank. 2004;5:129-134.
15. Becchetti E, Pagliarini A, Evangelisti R, Negri P. Composizione in glicosaminoglicani della membrana amnio-coriale umana in condizioni normali e patologiche. Boll Soc It Biol Sper. 1981;57:829-834.
16. Matthews RN, Bennett JP, Faulk WP. Amnion on wounds: A perspective. Reprod Immunol. 1981:269-280.
17. Gajiwala AL, Sharma V. Use of irradiated amnion as a biological dressing in the treatment of radiation induced ulcers. Cell Tissue Bank. 2003;4:147-150.
18. Somerville PG. L’utilisation de la membrane amniotique comme pansement biologique dans le traitement des ulcères chroniques de jambe. Phlébologie. 1982;35:223-229.
19. Gajiwala K, Gajiwala AL. Evaluation of lyophilised, gamma-irradiated amnion as a biological dressing. Cell Tissue Bank. 2004;5:73-80.
20. Toda A, Okabe M, Yoshida T, Nikaido T. The potential of amniotic membrane/Amnion-derived cells for regeneration of various tissues. J Pharmacol Sci. 2007;105:215-228.
21. Marvin KW, Keelan JA, Eykholt RL, Sato TA, Mitchell MD. Expression of angiogenic and neurotrophic factors in the human amnion and choriodecidua. Am J Obstet Gynecol. 2002;187:728-734.
22. Gris O, Wolley-Dod C, Güell J.L, et al. Histologic findings after amniotic membrane graft in the human cornea. Ophthalmology. 2002;109:508-512.
23. Quinby WC, Hoover HC, Scheflan M, Walters PT, Slavin SA, Bondoc CC. Clinical trials of amniotic membranes in burn wound care. Plast Reconstr Surg. 1982;711-716.
24. Egan TJ, O’Driscoll J, Thakar DR. Human amnion in the management of chronic ulceration of the lower limb: a clinico-pathologic study. Angiology. 1983:197-203.
25. Ward DJ, Bennett JP, Burgos H, Fabre J. The healing of chronic venous leg ulcers with prepared human amnion. Br J Plast Surg. 1989;42:463-467.
26. Ward DJ, Bennett JP. The long-term results of the use of human amnion in the treatment of leg ulcers. Br J Plast Surg. 1984;37:191-193.
27. Hernandez Cañete CM. La membrana amniocorial en el tratamiento de las ulceras vasculares. Angiologia. 1993;3:112-116.
28. Shun A, Ramsey-Stewart G. Human amnion in the treatment of chronic ulceration of the leg. Med J Aust. 1983;2:279-283.
29. Pesteil F, Oujaou-Faïz K, Drouet M, et al. Utilisation des membranes amniotiques cryopréservées dans la prise en charge des ulcères vasculaires rebelles. J Mal Vasc. 2007;32:201-209.
30. Miki T, Strom SC. Amnion-derived pluripotent/multipotent stem cells. Stem Cell Rev. 2006;2:133-141.
31. Parolini O, Alviano F, Bagnara GP, et al. Concise review: Isolation and characterization of cells from human term placenta: outcome of the first International Workshop on placenta derived stem cells. Stem Cells. 2008;26:300-311.
32. Sippel KC, Ma JJ, Foster CS. Amniotic membrane surgery. Curr Opin Ophthalmol. 2001;12:269-281.
