Artificial venous valves: an ongoing quest to treat end-stage deep venous insufficiency
Indianapolis, USA
ABSTRACT
End-stage deep venous insufficiency is unrelenting venous hypertension with sequelae, and no standard option is available, or all options have been tried and found wanting. In such cases, there is an opportunity for an artificial venous valve to be used as a native valve. For decades, substitute valves have been studied experimentally, raising hope of bench-to-bedside transfer. This quest is reviewed with an emphasis on current clinical practice. Venous valves have been made entirely of non-autologous tissues: synthetics, xenografts, or allografts. Many have failed in early experimental evaluation, with some advancing to the clinical arena, but few remain in research and development. Valves constructed from autogenous cells, or from autogenous venous tissue, not originally “de novo” valve tissue, have proven more promising. A variety of techniques have been used clinically, and improved venous hemodynamics and valve competency have been demonstrated. However, the majority of these valve studies await confirmation by other investigators over extended periods.
INTRODUCTION
It is apparent from the current literature that venous ulceration will recur even after the most aggressive treatment of superficial and perforator disease in patients with clinical class C5,6 disease.1 Ulcer recurrence is more common in patients with postthrombotic deep venous insufficiency (~ 70%), but is also seen in patients with primary deep venous insufficiency (~ 30%). The role of proximal iliac vein obstruction may be more prominent than once expected,2 but surgery to correct deep venous insufficiency (DVI) remains an appropriate option in selected patients. Even in the best hands, and with an architecturally preserved venous valve, about one-third of internal valvuloplasty repairs will fail within 5 years.3,4 Those requiring valve transposition or transplantation procedures fare less well, with only about 30% to 40% of valves competent at 5 years.3-5 In the face of unrelenting symptomatic deep venous insufficiency, and the lack of a standard treatment option, there is an opportunity for the use of an artificial venous valve. The quest to address this need has been ongoing for decades, and many avenues have been explored.
The standard dictionary definition of “artificial” is “not arising from natural growth.” Therefore, and for this review, an artificial venous valve is not considered as a “de novo” venous valve. In general, two categories of artificial venous valves have been studied: valves devoid of autogenous components; and valves constructed, at least partially, from autogenous components.
REVIEW OF THE LITERATURE
NONAUTOGENOUS VALVES
Some investigations have never advanced past the point of a promising valve studied for hemodynamic responsiveness. In a lyophilized cadaveric vein, a valve acts mechanically much like a native valve when rehydrated.6 The cusps withstood greater than 350 mm Hg retrograde pressure without leakage, and the closure time was 0.31+ 0.03 seconds. No animal implants or clinical investigations have been reported.
Some valves tested in animal models fared quite poorly, and, therefore, further investigation was deemed unwarranted. Fresh allograft vein segments containing a valve were transplanted into the femoral vein of 14 dogs with only 7% patent at four weeks.7 A human umbilical vein fitted over an aluminum mandrel, sculptured into a bicuspid valve and then glutaraldehydefixed, was implanted as a xenograft. All ten canine transplants failed in three days.8 A liquid pellethane bicuspid valve was poured and fashioned using the same aluminum mandrel as that used for the umbilical vein experiment. All 10 canine implants thrombosed in 8 days.8
Animal studies, or occasionally even unrelated clinical studies, raised hope. Platinum or pyrite-carbon–covered, titanium, center-hinged bileaflet valves implanted in the dog femoral vein had 100% patency and competency at three months.9 Unfortunately, extensive neointimal overgrowth resulted in valve failure within 2 years.10 These results hold some promise that modifications might be able to extend valve life into a useful clinical range.
Decellularization of allograft veins containing valves could provide a transplant devoid of potentially immunogenic donor cells. A cryopreserved decellularized allograft, used as an arteriovenous fistula (AVF) for dialysis access, incited little antigenic response, with good overall function.11 When implanted into the right ventricular outflow tract, a relatively high flow situation, pulmonary valve allografts functioned well for at least 6 months in a sheep model.12 Implantation of pulmonary valve allografts as an adjunct to the Ross procedure did not induce an antibody response, as determined by panel reactive antibody (PRA) testing.12 However, decellularized vein–containing valve allografts, implanted as venous valves in recipient sheep, and unaided by supportive anticoagulation, all failed in six weeks.13 Although this animal study was unsuccessful, clinical experience, with the same material as an AVF or cardiac valve, suggests that further study might be rewarding.
There are valve substitutes free of autogenous tissue that have advanced to clinical trial. A single allograft valve utilizing standard allogenic cross-matching, and cryopreservation for storage, has reached this level of investigation. As a preamble to the clinical trial, dog erythrocyte antigen–matched and cryopreserved veins containing valve allografts were transplanted into recipient dogs with experimental hind limb venous insufficiency. Following ligation of a post-implant highflow dAVF at 3 to 6 weeks, all four transplants remained patent and competent for three more weeks, at which time sacrifice demonstrated acceptable histologic findings.14 The inner surface had an endothelial-like cell covering, and cusp sinuses were free of thrombus. A multicenter feasibility study (Figure 1) unfortunately suggested that a low-grade rejection phenomenon was damaging the allogenic femoral vein valves, with primary patency rate of 67% and primary competency rate of only 56%.15 A two-year clinical study reported a disappointing 27% patency and competency rate.16 The cryopreserved valve allograft failed in early and midterm clinical trial, and is not considered a suitable valve substitute for treating DVI.
Another clinical investigation utilized a cryopreserved allograft pulmonary valve monocusp implanted surgically into the common femoral vein in patients with longstanding, active venous ulcerations (> 3 years).17 It was difficult to determine if the patients had an autogenous alternative, but the technique is unique. Fourteen of 40 (33%) implants were incompetent at follow-up, the length of which was not clearly stated. If the valve remained competent, the clinical results were excellent (24/27 or 89%), while recurrent reflux led to recurrent ulceration or non-healing. Mention of an immunogenic reaction, especially without blood type– specific matching, was considered a problem. No further report has been forthcoming.
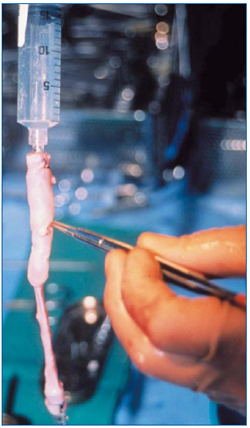
Figure 1. Photograph showing valve competence prior to implant of a cryopreserved vein-containing valve.
Cryopreserved femoral valved vein (cryovalve) is available from CryoLife (CryoLife, Inc. Kennesaw, Ga). During testing, the valve remains competent to at least 125 mm Hg of retrograde pressure. Data suggest that primary valvuloplasty may be required post-thaw to ensure initial competence.16 As noted previously; this valve fails quickly over time, but what if one could temper the chronic rejection that seems to damage the valve over time?15,16 Immunosuppressive agents would have to be well tolerated and not risk systemic infection. The suggested mechanism of rejection is the cytotoxic T cell response to foreign endothelium,18 which could be modified with agents such as aziathioprine or cyclosporin A. No clinical trials have tested this hypothesis.
Glutaraldehyde-preserved vein-containing valve allografts, with adjuvant distal AVF support, remained patent (80%), but rarely competent (25%), in a 7-week canine study.19 Glutaraldehyde-preserved bovine cardiac valves function well as human heart valves, and there is one report of a successful open surgical implantation of a glutaraldehyde-preserved pericardial allograft monocusp valve designed to treat a patient with DVI.20 This tissue is no longer available from the supplier, so the authors have changed their line of investigation.17 The technology existed to construct glutaraldehydepreserved bovine venous valves for clinical use, and therefore this became an area for investigation. Early hemodynamic testing of bovine jugular vein valves proved very promising, and the vein diameter was of appropriate size for human use.21 The possibility of valve transplantation by a percutaneous route was demonstrated experimentally and reported in the literature (Figure 2).22
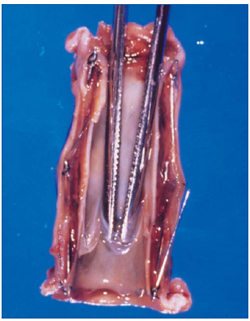
Figure 2. Photograph of a vein-containing valve that has been attached to a Z–stent and explanted after percutaneous implantation into a canine model. Note the normal appearance of the valve, highlighted by the instrument, with some narrowing at the proximal end of the stent.
Subsequently, in a swine model, a glutaraldehyde-preserved bovine vein-containing valve was implanted via a percutaneous route. In the three surviving animals, the valves were patent and competent.23 Further experimentation in an even larger animal study suggested that this approach was possible, but there was still some concern with valve competence, since while 5 of 5 valves at 4 weeks were patent, only 3 were competent.24 Additional unpublished data supported the concept that a clinical feasibility trial was appropriate. A report from the Jobst Institute confirmed that two percutaneous placements of this device were accomplished as a phase 1 trial.25 Recruitment was difficult due to the stringent criteria to insure that only end-stage patients were being treated. At approximately 1.3 years of follow-up, both stent valves were patent, but one was found to be incompetent at 14 months. These and other early results were somewhat discouraging, and pointed to a need to redesign. The new design likely failed to solve clinical concerns since the parent company no longer exists.
The most recent venture into the use of a nonautogenous valve for clinical use is a bioprosthetic valve made of porcine small intestinal submucosa (SIS). A bioprosthetic, bicuspid stent-based xenograft valve was developed and deployed percutaneously in the external jugular vein of a sheep model.26 SIS is essentially a collagen skeleton with growth factors, which was stretched over a square metal frame with a slit cut to form the valve opening. The valve was found to be resistant to thrombosis, and becomes repopulated with endothelial cells from the recipient.26,27 An 88% patency and competency rate was reported, but tilting led to valve malfunction or occlusion in 3 animals.26 Three patients were treated, thus demonstrating the feasibility of the approach.28 A design change enabled automatic centering of the valve, and 6 of 8 valves were competent at 5 weeks in an animal study.29 The company sponsoring the project (Cook, Inc., Bloomington, Ind) is continuing research and development, with early clinical studies performed outside the United States. A third design change is planned to improve venous valve hemodynamics and prevent cusp thickening.
AUTOGENOUS VALVES
Repopulating a decellularized valved vein allograft with donor smooth muscle and endothelial cells would make a transplant much like an autogenous valve, but with an allograft infrastructure. This hybrid is difficult to categorize, but, I believe, fits the autogenous category best for the purposes of this review. In a sheep model, such a seeded allograft was transplanted into the external jugular vein of the cell donor. Devoid of long-term anticoagulation, 9 of 12 seeded allografts were patent and competent at 12 weeks. One transplant had occluded, and two valves were frozen by neointimal ingrowth.13 These allografts did perform much better than unseeded allografts, which universally failed, but not as well as 8 autografts, which were all patent and competent. This is promising experimental work.
Intussusception of an autogenous vein forms a bicuspid valve by placing two sutures 180 degrees from each other to hold the intussusceptum in place.30-32 The tissue is autogenous, but the valve is artificially constructed. In canine studies performed without chronic anticoagulation, short-term patency was excellent, with valve competence demonstrated at physiologic pressures. The valve was, of course, thicker than a native valve by virtue of its method of construction (Figure 3).30 When transplanted into the femoral vein of a canine DVI model, the 90% venous refill time was modestly improved, but not the venous filling time, suggesting a less rapidly responsive valve when compared to a native valve.30 The British experience with this type of valve was evaluated by the Harvey strip test and descending venography, and long-term competence (1-112 days) was demonstrated in animals.31 All 41 valves were immediately competent by the strip test, 38 by descending venography, and 24 of 27 fully competent to a vertical pressure gradient of up to 250 cm H2O in this animal study.31 A modification to allow thinner valve cusps showed that the valve opened rapidly with minimal retrograde pressure, closed at a pressure of 3 to 5 cm of water, and could withstand physiologic hydrostatic pressure without reflux.32 In the absence of prolonged anticoagulation, a thin layer of thrombus formed along the thinned cusp walls, resulting in valve incompetence. Overall, with some modifications, this could function as a substitute valve, but animal studies have raised concerns as to optimal function and a higher-than-normal risk of thrombosis. Although an invaginated valve has been used in the saphenous system to prevent reflux,33 this valve design has not yet been investigated in the deep venous system of patients.
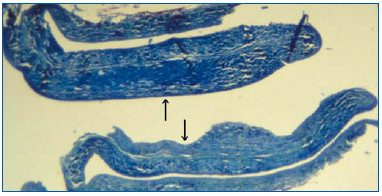
Figure 3. Photograph of an intussusception-type venous valve showing the double thickness of the valve construct, which may help to explain the slower response of the valve during hemodynamic study.
There are clinical reports of attempts to use autogenous venous tissue to fashion intraluminal venous valve cusps. It is my belief that this avenue of study initially resulted from the pressing need for a valve during surgery, when no other option was available.
Raju and Hardy report a small series of de novo valve reconstruction procedures.34 Using autogenous vein from various locations, and after trimming adventitia and part of the media, semilunar cusps were cut to shape and sutured into the recipient vein. The non-endothelial surface was directed toward the lumen to decrease the risk of thrombosis.34 All 7 valves were patent at 15 to 24 months of follow-up, primary healing of venous ulcers was recorded in 6 patients, with one requiring a skin graft to complete the healing process. No recurrences were noted.
Plagnol et al invaginated a stump of the great saphenous vein into the femoral vein to make a bicuspid valve, and 19 of 20 clinical reconstructions were patent and competent at a mean of ten months.35 Reflux was noted in one case because of insufficient valve length. I have some concerns regarding invagination of an adventitial surface into the venous lumen, but these are not substantiated in this report.
Maleti made bicuspid or monocusp venous valves (Figure 4) by dissecting an inner layer from the thickened post-phlebitic vein wall to form the cusp(s). The initial 7 cases were sufficiently successful to warrant further study.36 At the 2005 American Venous Forum, Lugle and Maleti reported the construction of 18 venous valves in 16 patients with recurrent or non-healing venous ulcers.37 Six months of chronic anticoagulation was standard. At an average of 22 months, 83% of the valves remained primarily patent, with improved duplex and air plethysmographic findings. Early thrombosis below the valve was observed in two patients, and one patient experienced a late occlusion after beginning oral contraceptives. Corcos et al report one case in which the “intimal flap” method of constructing a venous valve was successful in healing a venous ulcer, and in improving venous hemodynamics.38
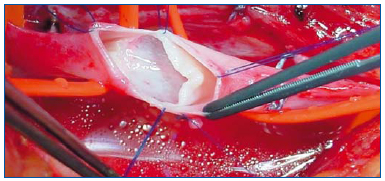
Figure 4. Photograph of a Maleti-type monocusp venous valve constructed by dissecting the inner wall of a post-phlebitic vein away from the outer wall to form a venous cusp. This photograph was generously provided by Dr. Oscar Maleti with permission to publish.
The early and midterm results are certainly promising for all the reported autogenous vein methods of new valve construction, but few methods have been substantiated by other investigators.
CONCLUSIONS
Valve cusps made of autogenous vein are currently the only artificial venous valves available with at least preliminary data to support their use in cases of chronic deep venous insufficiency, for which there are no standard options. Non-autogenous, off-the-shelf venous valve substitutes are in research and development, or have hitherto failed clinical evaluation. With modifications, some of the latter do hold promise for the future.

REFERENCES
2. Neglen P, Thrasher TL, Raju S. Venous outflow obstruction: an underestimated contributor to chronic venous disease. J Vasc Surg. 2003;38:879-885.
3. Masuda EM, Kistner RL. Long-term results of venous valve reconstruction: a four to twenty-one year follow-up. J Vasc Surg. 1994;19:391-403.
4. Perrin M. Reconstructive surgery for deep venous reflux: a report on 144 cases. Cardiovascular Surg. 2000;8:246-255.
5. Sottiurai VS. Results of deep-vein reconstruction. Vasc Surg. 1997;31:276- 278.
6. Reeves TR, Cezeaux JL, Sackman JE. Mechanical characteristics of lyophilized human saphenous vein valves. J Vasc Surg. 1997;26:823-828.
7. McLachlin AD, Carroll SE, Meads GE, Amecher AL. Valve replacement in the recanalized incompetent superficial femoral vein in dogs. Ann Surg. 1965;162:446-452.
8. Hill R, Schmidt S, Evancho M, et al. Development of a prosthetic venous valve. J Biomed Mater Res. 1985;19:827-832.
9. Taheri SA, Rigan D, Wels P, et al. Experimental prosthetic vein valve. Am J Surg. 1988;156:111-114.
10. Taheri SA, Schultz RO. Experimental prosthetic vein valve. Long-term results. Angiology. 1995;46:299-303.
11. Madden R, Lipkowitz G, Benedetto B, et al. Decellularized cadaver vein allografts used for hemodialysis access do not cause allosensitization or preclude kidney transplantation. Am J Kidney Dis. 2002;40:1240-1243.
12. Elkins RC, Dawson PE, Goldstein S, et al. Decellularized human valve allografts. Ann Thorac Surg. 2001;71:S428-S432.
13. Teebken OE, Puschman C, Aper T, Haverich A, Mertsching H. Tissueengineered bioprosthetic venous valve: a long-term study in sheep. Eur J Vasc Endovasc Surg. 2003;25:305-312.
14. Burkhart HM, Fath SW, Dalsing MC, et al. Experimental repair of venous valvular insufficiency using a cryopreserved venous valve allograft aided by a distal arteriovenous fistula. J Vasc Surg. 1997;26:817-822.
15. Dalsing MC, Raju S, Wakefield TW, Taheri S. A multicenter, phase I evaluation of cryopreserved venous valve allografts for the treatment of chronic deep venous insufficiency. J Vasc Surg. 1999;30:854-866.
16. Neglen P, Raju S. Venous reflux repair with cryopreserved vein valves. J Vasc Surg. 2003;37:552-557.
17. Garcia-Rinaldi R, Soltero E, Gaviria J, et al. Implantation of cryopreserved allograft pulmonary monocusp patch to treat nonthrombotic femoral vein incompetence. Tex Heart Inst J. 2002;29:92-99.
18. Carpenter JP, Tomaszewski JE. Human saphenous vein allograft bypass grafts: immune response. J Vasc Surg. 1998;27:492-499.
19. Kaya M, Grogan JB, Lentz D, et al. Glutaraldehyde-preserved venous valve transplantation in the dog. J Surg Res. 1988;45:294-297.
20. Garcia-Rinaldi R, Revuelta JM, Martinez MJ, et al. Femoral vein valve incompetence: treatment with a xenograft monocusp patch. J Vasc Surg. 1986;3:932- 935.
21. DeLaria GA, Phifer T, Roy J, et al. Hemodynamic evaluation of a bioprosthetic venous prosthesis. J Vasc Surg. 1993;18:577-586.
22. Dalsing MC, Sawchuk AP, Lalka SG, Cikrit DF. An early experience with endovascular venous valve transplantation. J Vasc Surg. 1996;24:903-905.
23. Gomez-Jorge J, Venbrux AC, Magee C. Percutaneous deployment of a valved bovine jugular vein in the swine venous system: a potential treatment for venous insufficiency. J Vasc Interv Radiol. 2000;11:931-936.
24. Teijink JA, Patterson M, Quijano TC, Moll FL. A percutaneous approach to deep venous valve insufficiency with a new selfexpanding venous frame valve. J Endovasc Therapy. 2003;10:341-349.
25. Gale SS, Shuman S, Beebe HG, et al. Percutaneous venous valve bioprosthesis: initial observations. Vasc & Endovasc Surg. 2004;38:221-224.
26. Pavcnik D, Uchida BT, Timmermans HA, et al. Percutaneous bioprosthetic venous valve: a long-term study in sheep. J Vasc Surg. 2002;35:598-602.
27. Brountzos E, Pavcnik D, Timmersmans HA, et al. Remodeling of suspended small intestinal submucosa venous valve: an experimental study in sheep to assess the host cells’ origin. J Vasc Interv Radiol. 2003;14:349-356.
28. Pavcnik D, Machan L, Uchida B, et al. Percutaneous prosthetic venous valves: current state and possible applications. Techniques in Vascular & Interventional Radiology. 2003;6:137-142.
29. Pavcnik D, Kaufman J, Uchida B, et al. Second-generation percutaneous bioprosthetic valve: a short-term study in sheep. J Vasc Surg. 2004;40:1223-1227.
30. Dalsing MC, Lalka SG, Unthank JL, et al. Venous valvular insufficiency: influence of a single venous valve (native and experimental). J Vasc Surg. 1991;14:576- 587.
31. Wilson NM, Rutt DL, Browse NL. In situ venous valve construction. Br J Surg. 1991;78:595-600.
32. Rosenbloom MS, Schuler JJ, Bishara RA, et al. Early experimental experience with a surgically created, totally autogenous venous valve: a preliminary report. J Vasc Surg. 1988;7:642-646.
33. Burnette WJ. Construction of venous valves. Am Surg. 1954;20:499-505.
34. Raju S, Hardy JD. Technical options in venous valve reconstruction. Am J Surg. 1997;173:301-307.
35. Plagnol P, Ciostek P, Grimaud JP, Prokopowicz SC. Autogenous valve reconstruction technique for postthrombotic reflux. Ann Vasc Surg. 1999;13:339-342.
36. Maleti O. Venous valvular reconstruction in postthrombotic syndrome. A new technique. Journal des Maladies Vasculaires. 2002;27:218-221.
37. Lugle M, Maleti O. Neovalve construction in postthrombotic syndrome. Presented at the American Venous Forum, 17th Annual Meeting; February 9-13, 2005; San Diego, Calif.
38. Corcos L, Peruzzi G, Procacci T, et al. A new autologous venous valve by intimal flap. One case report. Minerva Cardioangiologica. 2003;51:395-404.
