Erysipelas and lymphedema
Tours, France
SUMMARY
Erysipelas is a nonnecrotizing bacterial hypodermal cellulitis usually associated with streptococcal infection. It may be a mainly secondary complication of chronic lymphedema, and occurs in 20% to 30% of cases. The first presenting signs are sudden fever and shivering. The clinical feature is inflammatory plaque, which is often chronic and accompanied by fever. Inflammatory plaque is promoted by lymph stasis, and is marked by inflammatory episodes that often regress spontaneously. Erysipelas per se is mainly treated with antibiotics, and adjuvant therapies are not justified. The prevention of recurrence is primary. Since lymphedema is the first risk factor for recurrence, its treatment and risk of occurrence must be considered. This includes physiotherapy, well-adapted compression therapy, and avoidance of wounds.
INTRODUCTION
The lymphatic system has a role in antigen presentation and defense against infection. Infection is the most common type of complication observed in lymphedema, and is promoted by lymphatic system dysfunction, which causes locoregional immune disorders. Infectious complications are primarily bacterial, and most commonly are erysipelas (cellulitis) and sometimes lymphangitis. ß-hemolytic streptococcus (groups A, C, G) is the organism usually (and even almost exclusively) observed in everyday practice. Bacterial complications are promoted by the abundance of proteins characteristic of edema in lymphatic insufficiency related to obstructed lymphatic vessels. This high protein content of the interstitial fluid is an ideal culture medium for the growth of bacteria.1
ERYSIPELAS: DIAGNOSIS
Erysipelas is a nonnecrotizing dermo-hypodermal bacterial infection without involvement of the superficial aponeurosis.1 Erysipelas (also known as cellulitis in English-speaking countries) is the clinical presentation most often observed in lymphedema, which it complicates in 20% to 30% of cases.1 It occurs slightly more frequently in secondary lymphedema than in primary lymphedema.
The diagnosis of erysipelas is clinical2
Erysipelas is often of sudden onset, marked by frank systemic signs—fever >38°5, chills—and general malaise. Patients with lymphedema, who present with recurrent episodes of erysipelas, often recognize these inaugural signs. Local signs develop within a few hours: a red, warm, painful, inflammatory spreading lesion, with centrifugal extension within a few days. Erysipelas can start at any point of lymphedema and can extend to all or part of the lymphedematous cutaneous tissue, in an anterograde or retrograde pattern. Inflammatory, satellite adenopathy and lymphangitis are associated with erysipelas in 25% to 50% of cases.3
In the case of erysipelas complicating lymphedema, the clinical presentation is more serious. Statistically significant differences (P<0.05) were seen between 20 controls with erysipelas but without lymphedema and 10 age- and sex-matched patients with lymphedema hospitalized for erysipelas: prolonged persistence of fever, more frequent tachycardia, delayed recovery, and positive blood cultures (30% vs 0%).4
The diagnosis of erysipelas is clinical: sudden occurrence of an inflammatory lesion that spreads within a few days, preceded by or concomitant with fever and chills, and general malaise. No bacterium other than ß-hemolytic streptococcus has been demonstrated as responsible for erysipelas.2 Streptococcus was isolated 15 times more often in lymphedema with an infectious complication than outside any acute inflammatory episode, and serology testing for streptococcus (ASLO) was more often positive in patients with lymphedema (78%) than in a healthy control population (46%).1 Bacteriological samples are positive in erysipelas in only 4% to 35% of cases with standard methods. Using the most sophisticated methods (immunofluorescence, polymerase chain reaction), streptococcus is isolated at a frequency of 70% to 80%.5 Other bacteria (Staphylococcus aureus, Enterobacteriaceae, Pseudomonas) have been isolated alone or in combination with streptococcus. But to date a causal relationship has never been demonstrated.
Furthermore, these bacteria are commonly found on skin and colonize wounds, and their isolation from samples collected from the skin is difficult to interpret. Laboratory tests are not helpful in establishing the diagnosis: the complete blood count shows leukocytosis in one-half of cases, and a nonspecific inflammatory syndrome is indicated by laboratory findings in two-thirds of cases.3
Causes of febrile inflammatory spreading lesions other than erysipelas
Erysipelas should be differentiated from other infections sometimes observed in lymphedema, such as lymphangitis, most often streptococcus-related (rarely staphylococcal), or necrotizing fasciitis (most often streptococcal).
Lymphangitis is characterized by the occurrence of an inflammatory streak (red, warm, and painful) whose topography is that of the superficial lymphatic vessels (which themselves are satellites of the superficial venous system). It is accompanied by fever. There is no inflammatory spreading lesion.
Necrotizing dermo-hypodermal bacterial infection or necrotizing fasciitis is characterized by necrosis of the aponeurosis and myositis, resulting in a presentation of infectious gangrene. Diffuse, indurated edema extends beyond the margins of the erythematous and sometimes slightly inflammatory spreading lesion. Deep necrosis may be manifest in the initial stage solely as a cyanotic, grayishblue, poorly demarcated swelling, with a geographical map-like presentation. Fever is a usual finding but can be mild or absent. A septic syndrome (with hemodynamic signs, hypoxia, and thrombocytopenia) develops subsequently. In lymphedema, subacute forms of necrotizing dermo-hypodermal bacterial infection with superficial necrosis are observed; these are absent in typical erysipelas. This should prompt emergency hospitalization of the patient. In a patient with lymphedema, it is important to differentiate necrotizing fasciitis, which is rarely observed, from erysipelas, since systematic surgery should be avoided in all cases of atypical erysipelas whose course is not immediately favorable because healing is difficult to obtain. Necrotizing fasciitis is a life-threatening medical-surgical emergency and must be treated in a specialized hospital department.
In lymphedema, chronic, inflammatory spreading lesions with little or no fever are often seen. They correspond to skin inflammation mediated by macrophages, lymphocytes, and cytokines,6 without bacterial infection. Such inflammatory spreading lesions are favored by lymph stasis and are similar to findings in stasis dermatitis observed in chronic edema. Their course is marked by inflammatory episodes that often regress spontaneously. In spite of their chronic nature and the absence of a frank infectious syndrome, it is usually difficult to eliminate the role of streptococcus in the case of such inflammatory spreading lesions. Their assumed pathophysiology in lymphedema (major secretion of cytokines subsequent to lymph stasis) has been the rationale for trials of oral steroid therapy. This treatment produces a prompt decrease in signs of inflammation. But such an approach cannot be recommended in practice because it is potentially hazardous. Cases of necrotizing fasciitis induced or exacerbated by anti-inflammatory drugs have been reported.
Febrile dermo-hypodermal lesions of a surgical scar (in particular breast scars or after breast implant surgery) have been reported,2 and may be infectious (erysipelas) or allergic (justifying steroid therapy).
Other acute forms of dermo-hypodermal bacterial infection are caused by Erysipelotrix rhusiopathiae (Rouget’s swine erysipelas), Haemophilus influenzae, Pasteurella multocida, and Borrelia borgdorferi. They have a different medical history or a less inflammatory, less febrile clinical presentation without the typical chronology (systemic syndrome followed by an inflammatory spreading lesion a few hours later), or both.
In summary, the diagnosis of erysipelas is solely clinical, and does not require any laboratory tests, in particular, bacteriological.
LYMPHEDEMA AND ERYSIPELAS: MANAGEMENT
Lymphedema is by far the most important risk factor for erysipelas (relative risk: 71.2) (Table I).7 The risk of recurrence of erysipelas after a first episode in patients with lymphedema is very high (greater than 50% within a year of the first episode), and increases as recurrences occur.
Treatment of erysipelas
The reference treatment of erysipelas is pristinamycin at a dosage of 3 g/d for two weeks.8 In a randomized, controlled study of 289 adults with erysipelas, the cure rate was 81% vs 67% for treatment with intravenous penicillin followed by a switch to oral penicillin (per protocol analysis, a statistically significant difference).8 In the absence of pristinamycin, treatment with oral amoxicillin (3-4.5 g/d, in 3 daily doses) for two weeks has been proposed.9
Oral therapy allows outpatient treatment, but hospitalization is necessary if:
1.there is doubt regarding the diagnosis, or there are serious systemic signs, comorbid conditions, or a social context which makes home treatment of the patient impossible, in which case admission should be immediate;
2. fever persists after more than 72 hours of treatment or new local or locoregional signs recur or both.10 In such cases, treatment with intravenous penicillin G (10-20 million units in 4-6 infusions a day) is the reference treatment.
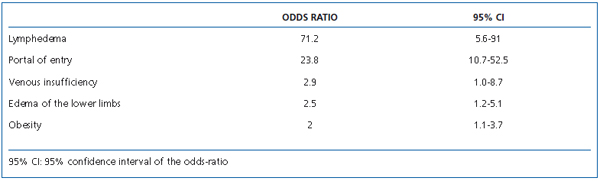
Table I. Erysipelas: risk factors.7
The value of adjuvant therapies was analyzed at a consensus conference organized by the French Society of Dermatology, the French Society of Lymphatic Disease, and the French Society of Infectious Diseases.9 Local, antiseptic, or antibiotic therapy is not necessary. Systematic anticoagulant prophylaxis is not justified if lymphedema is complicated by erysipelas alone. In fact, the risk of deep vein thrombosis (DVT) is only 0.7%- 4.5%.11 There is no rationale for systematic screening for DVT with duplex scanning.
Treatment of lymphedema
No study has demonstrated that physiotherapy or compression promotes bacterial infection in lymphedema. In a few isolated cases, pressure therapy has been implicated as having induced episodes of lymphangitis or erysipelas. This assertion has not been confirmed by a large retrospective study.12 In practice, it may be advisable to avoid manual exercises to promote lymphatic drainage and pressure therapy in the case of erysipelas, and to resume such measures only two to four weeks after the start of antibiotic therapy. Compression therapy is not contraindicated in erysipelas.11 A consensus conference has recommended the evaluation of compression therapy in reducing edema and the risk of venous thrombosis.9 From a pragmatic standpoint, considering the events in inflammation, it is advisable to avoid vigorous compression therapy during the first two weeks of treatment.
Physical therapy, even intensive (decongestive complex physiotherapy), does not justify the use of streptococcal prophylaxis. On the other hand, in the case of surgery (venous lymphatic anastomosis, etc) antibiotic prophylaxis should be considered in patients with a history of lymphedema complicated by infection.
Treatment of factors that promote erysipelas
Treatment should minimize local risk factors for lymphedema (Tables I and II). The main portals of entry of erysipelas are intertrigo and wounds.7
Wound disinfection with an antiseptic (chlorhexidine or Betadine®) is recommended, although it has not been validated and the efficacy of antiseptics on abraded skin has recently been called into question.13 If a wound is infected (locoregional signs of inflammation), the best treatment is a topical antibiotic such as Fucidin®.14 If systemic signs of infection are present, oral antibiotic therapy with pristinamycin as in erysipelas is the best validated treatment. Screening and treatment of a fungal infection, in particular, between the toes, is the most effective measure in terms of prevention of infectious complications, considering the incidence of this disorder.6 Treatment of such fungal infection is based on use of an imidazole cream in a single application (Fonx®, Amycor®), and the use of imidazole powder (Daktarin®, Pevaryl®) in the patient’s shoes and socks. If recurrence is frequent, long-term prophylaxis can be recommended. Use of an antifungal cream and appropriate skin care decrease the recurrence of erysipelas.15
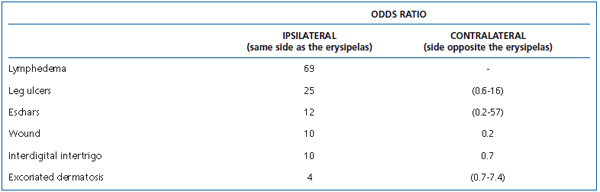
Table II. Erysipelas: risk factors (local or systemic).7
Physiotherapy for lymphedema decreases the recurrence rate.16,17 In a study of 20 patients, the wearing of elastic compression stockings after a first episode of erysipelas reduced the risk of recurrence 5-fold (results at 5 years: 5% recurrence vs 25% in the absence of treatment).16 Identical results have been reported with physiotherapy combined with decongestive therapy.17
Prophylaxis of recurrence of erysipelas
When factors that promote erysipelas are difficult to control, and erysipelas or lymphangitis has recurred in spite of good management of lymphedema, streptococcal prophylaxis is necessary.18 It should be of long duration or even permanent since the effect of treatment is only temporary.9 It requires use of penicillin: intramuscular benzathine penicillin 1.2-2.4 million units every 2-3 weeks, or oral penicillin V 2-4 million units in 2-3 doses a day. The intramuscular route, which is painful, ensures better compliance and has proven effective.19 In this study, treatment with benzathine penicillin for 1 year reduced acute episodes of erysipelas (results at 1 year: 0.5 vs 4.6 before treatment), and at 2 years, ie, 1 year after discontinuation of treatment, the reduction was still significant (results at 2 years: 1.9 vs 4.6 before treatment).19 In the case of ß-lactam allergy, it is advisable to prescribe a macrolide; roxithromycin (Rulid®) has been validated in erysipelas.18
CONCLUSION
Erysipelas is a frequent complication of lymphedema. It is caused by a ß-hemolytic streptococcus, which can destroy lymph vessels. Thus, secondary and tertiary prevention of lymphedema absolutely requires early management and prophylaxis of bacterial and fungal infections. Conversely, proper management of lymphedema, with reduction of the extent of lymphedema and daily care of the skin and integument, decreases the risk of bacterial and fungal infections.

REFERENCES
2. Vaillant L. Critères diagnostiques de l’érysipèle. Ann Dermatol Venereol. 2001;128:326-333.
3. Lorette G, Py F, Machet L, Vaillant L. Erysipèle : étude rétrospective de 100 cas. Med Hyg. 1990;48:764-768.
4. Woo PC, Lum PN, Wong SS, et al. Cellulitis complicating lymphedema. Eur J Clin Microbiol Infect Dis. 2000;4:294-297.
5. Bernard P, Bedane C, Mounier M, et al. Streptococcal cause of erysipelas and cellulitis in adults study using a direct immunofluorescence technique. Arch Dermatol. 1989;125:779-782.
6. Olszewski WL. Episodic dermatolymphangioadenitis (DLA) in patients with lymphedema of lower extremities before and after administration of benzathine penicillin preliminary study. Lymphology. 1996;29:126-131.
7. Dupuy A, Benchikhi H, Roujeau JC, et al. Risk factors for erysipelas of the leg (cellulitis): case-control study. BMJ. 1999;318:1591-1954.
8. Bernard P, Chosidow O, Vaillant L. French Erysipelas Study Group. Oral pristinamycin versus standard penicillin regimen to treat erysipelas in adults: randomised, non-inferiority, open trial. BMJ. 2002;325:864-868.
9. Prise en charge des érysipèles et fasciites nécrosantes. Conférence de consensus. Ann Dermatol Venereol. 2000;127:336-340.
10. Bernard P. Critères d’hospitalisation primaire et secondaire. Ann Dermatol Venereol. 2001;128:363-367.
11. Perrot JL, Perrot S, Laporte Simitsidis S. Un traitement anticoagulant est-il utile dans le traitement des érysipèles ? Ann Dermatol Venereol. 2001;128:352-357.
12. Vaillant L, Titon JP, Gaisne E, Barsotti J. Compression for lymphedema and cellulitis. J Am Acad Dermatol. 1990;23:952- 953.
13. Wolkenstein P, Vaillant L. Antiseptiques en peau lésée. Ann Dermatol Venereol. 1996;123:343-348.
14. Machet L, Wolkenstein P, Vaillant L. Antibiotiques topiques en dermatologie : efficacité, indications et effets secondaires. Ann Dermatol Venereol. 2000;127:425-431.
15. Shenoy RK, Kumaraswani V, Suma TK, et al. A double-blind, placebo-controlled study of the efficacy of oral penicillin, diethylcarbamazine or local treatment of the affected limb in preventing acute adenolymphangitis in lymphedema caused by bugian filariasis. Ann Trop Med Parasitol. 1999;1933:367-377.
16. Yasuhara H, Shigematsu H, Muto T. A study of the advantages of elastic stockings for leg lymphedema. Int Angiol. 1996;15:272-277.
17. Foldi E. Prevention of dermatolymphangioadenitis by combined physiotherapy of the swollen arm after treatment for breast cancer. Lymphology. 1996;29:48-49.
18. Bernard P. Dermo-hypodermal bacterial infections. Current concepts. Eur J Med. 1992;1:97-104.
19. Badger C, Seers K, Preston N, Mortimer P. Antibiotics/anti-inflammatories for reducing acute inflammatory episodes in lymphedema of the limbs. Cochrane Database Syst Rev. 2004;2:CD003143.
