Lymphatic system dysfunction in pediatric populations
Christoph M. PAPENDIECK
SUMMARY
Lymphatic system dysfunction in children has many presentations and syndromes and may affect all parts of the body. It can be classified according to the type of abnormality present as dysplasia of the lymphatics (LAD I), dysplasia of the lymph nodes (LAD II), or dysplasia of the lymphatics and lymph nodes (LAAD). Lymphatic dysplasias can be clinically represented by peripheral lymphedema alone or be associated with more and complex dysfunction in different locations, eg, intestinal lymph vessels, thoracic duct, and others. Primary lymphedema has for many years been the least understood form of lymphedema, but exploration of the underlying genetic causes of certain types of primary lymphedema is helping researchers identify and understand previously unrecognized syndromes. Carefully searching for lymphatic dysplasia in these patients, and if indicated in their relatives, as well as establishing the exact nature of the lymphatic dysplasia means that most cases of lymphatic dysfunction can now be diagnosed. This paper summarizes current knowledge on the recognition and classification of lymphatic system dysfunction in pediatric populations, with a focus on primary lymphedema.
INTRODUCTION
A broad spectrum of inherited and acquired diseases is characterized by an impaired ability of the lymphatic system to collect and transport fluid. The most easily recognizable feature of lymphatic vessel incompetence is the presence of tissue swelling, lymphedema, which arises as a consequence of insufficient lymph transport. It is important for healthcare practitioners to be aware of signs and symptoms that may be precursors to the clinical diagnosis of lymphedema as not only is the efficient drainage of interstitial fluid from the capillary beds essential to prevent edema, but it also allows pathogenic material, from infections and injuries, to pass into the lymphatic system where it can be effectively trapped. Earlier detection and treatment of lymphedema means that more can be done to prevent its progression.
STRUCTURE AND
FUNCTION OF THE LYMPHATIC SYSTEM
Vertebrates have developed a sophisticated system of recognition, rescue, integration, and transport of macromolecules and pathogenic material suspended in water. Located between the interstitial space and the deep jugular-subclavian venous system, this multipurpose system links the interstitial and intravascular space, returning fluids, solutes, and proteins filtered from the capillaries back to the systemic circulation. It has three major functions: removal of tissue fluid, production of immune cells, and absorption and transport of fatty acids and lipid-soluble vitamins to the circulatory system. To achieve this task the lymphatic system has a complex network of vessels throughout the body. In two clearly defined circuits, lymphatic channels from the upper right side of the body converge to form the right lymphatic duct, which empties into the confluence of the right internal jugular and subclavian vein, and lymphatic channels from the remainder of the body drain via Pecquet’s cistern (cisterna chyli) into the thoracic duct, which empties into the left internal jugular vein at its confluence with the left subclavian vein.1
All tissues of the body are continuously bathed in interstitial fluid. As the blood supplies nutrients and important metabolites to cells via the interstitial fluid and collects cellular waste products, the composition of the interstitial fluid continually changes. Approximately 90% of the interstitial fluid returns to the blood via the venous capillaries, but the remaining fluid, which also contains larger protein molecules unable to diffuse across the venous capillary walls as well as white blood cells, dead cells, bacterial debris, infected substances, enters the lymphatic system.1 The loss of this fluid (in adults, approximately 1-2 liters/day) would rapidly become lifethreatening if the lymphatic system did not function properly.
The smallest lymphatic vessels are the lymph capillaries, which begin in the tissue spaces as pre-capillaries.2 The wall of the pre-lymph capillary is composed of a single layer of overlapping endothelial cells with no basal membrane. When interstitial fluid volume and pressure increase, largely as a result of hydrostatic and colloidal osmotic pressure gradients, the space expands and the overlapping borders of the endothelial cells separate to allow unidirectional flow of fluid into the lymphatic capillaries and prevent backflow of lymph into the interstitial space.3 The lymphatic capillaries do not have valves, so lymph flows in the direction of lower pressure. This process creates a flow gradient for lymph from the interstitium into the lower pressure lymphatic capillaries and subsequently into the larger precollector and collector lymph vessels. Its functional unit is the lymphangion, surrounded by a spiral of muscle, which contracts when the lymphangion has expanded to a certain level, pumping the lymph from one lymphangion to the next. In addition, a system of one-way valves only allows the lymph to flow in one direction. Unlike the cardiovascular system, the lymphatic system is not closed and has no central pump. Lymph movement occurs despite low pressure due to the action of peristalsis, valves, and compression during contraction of adjacent skeletal muscle.
The lymphatic system plays an integral role in the immune functions of the body and is the first line of defense against disease. Approximately 500-600 lymph nodes occur along the entire length of the lymphatic system and are organized in groups that drain specific regions of the body.1 Their primary role is to filter, concentrate, and purify the lymph fluid. The nodes produce macrophages and lymphocytes, and cytokines, which form part of the body’s immune defense system. Lymphocytes are produced by stem cells in the bone marrow and then migrate to either the thymus or bone marrow (the primary lymphoid organs) where they mature into T-lymphocytes or B-lymphocytes, respectively. After maturation, both B- and T-lymphocytes circulate in the lymph and accumulate in secondary lymphoid organs including the splenic parenchyma, lymph nodes, and accessory lymphoid tissue (including the tonsils, adenoids, and Peyer’s patches).
The lymphatic system is also involved in the absorption and transport of fat to the circulatory system.1 Lymph vessels in the lining of the gastrointestinal tract absorb fats from food. Lymph draining from intestinal cells appears milky because of the fat globules that have been absorbed and is known as chyle. A malfunction of this part of the lymphatic system can result in serious malnutrition as a result of protein-losing enteropathy.
THE ETIOLOGY OF LYMPHEDEMA
An excess of interstitial fluid does not necessarily mean a malfunction of the lymphatic system as it is designed to cope with varying loads. Lymphedema only occurs when there is failure of lymph drainage as a result of lymph transport capacity falling below normal. In this situation, the normal volume of interstitial fluid formation exceeds the rate of lymphatic return, resulting in the accumulation of high-molecular-weight proteins in the interstitium. The high oncotic pressure in the interstitium favors the accumulation of additional water, which exacerbates the lymphedema. Eventually cell damage occurs because of the increase in interstitial pressure, disturbed cell nutrition, and transport of metabolites. Lymphedema of the extremities is clinically evident, but it is less widely recognized that lymphedema may also result in intracellular edema causing parenchymal cell lesions, which may appear in any organ drained by the lymphatics (hydrops). The two main classifications of lymphedema are primary and secondary.
Primary lymphedema
Primary lymphedema is due to an inherent defect within the lymphatic tissue itself and usually manifests as aplasia, hypoplasia, hyperplasia, and dysplasia of lymph tissue (lymph vessels and nodes), which results in the presence of abnormally high levels of proteins and tissue fluids in the interstitial space.4 Figures 1 to 4 illustrate examples of lower and upper limb lymphedema in infants. Primary lymphedema is usually further subdivided into three forms depending on age at presentation: congenital lymphedema (clinically evident at birth), lymphedema praecox (becomes clinically evident after birth, usually during puberty, and before age 35 years), and lymphedema tarda or Meige disease (becomes clinically evident after 35 years of age). These conditions often occur sporadically with no family history, and most frequently involve the lower extremities. All three forms of primary lymphedema are thought to originate from a developmental abnormality that is present, but not always clinically evident, at birth.
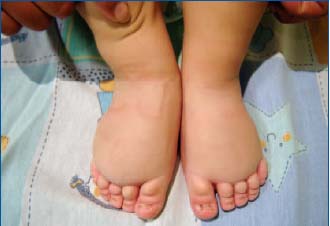
Figure 1. Primary lymphedema in both feet in an infant,
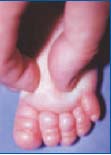
Figure 2. The Stemmer sign, and pitting
test. Newborn with primary
lymphedema.
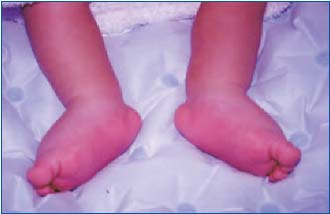
Figure 3. Primary lymphedema in both legs in an infant.
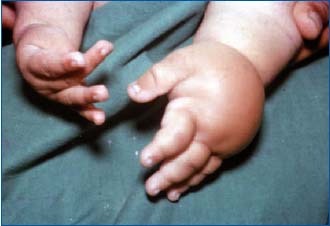
Figure 4. Primary lymphedema in one upper limb in an infant.
Secondary lymphedema
When the cause of lymphedema is attributed to trauma, infection, or surgical removal of lymph nodes, it is referred to as secondary or acquired lymphedema. In pediatric populations, secondary lymphedema is mainly caused by inflammatory or traumatic lymphatic injuries. Congenital causes include amniotic band (constriction band) syndrome,5 hair tourniquet syndrome,6 truncular venous hypertension because of intrinsic or extrinsic venous dysplasias or prenatal thrombosis. Obstructive mechanisms (pelvic tumor, compressive lymph flow disturbances) are less frequent than in the adult population. Noncongenital causes include parasitosis, drugs and pharmacological treatment, podoconiosis,7 and trauma. In the tropics, secondary lymphedema in children is most frequently caused by lymphatic filariasis, the most common cause of lymphedema worldwide, which affects an estimated 120 million people.8 The disease is caused by infection with the mosquito-borne parasites Wuchereria bancrofti, Brugia malayi, Mansonellas, and others. These live and reproduce in the lymphatic system causing progressive dysfunction and obstruction at the nodal level, resulting in lymphedema.9 Infection is concentrated in the tropics and does not account for the majority of cases of lymphedema seen in temperate zones. Other parasites causing infections that reside in the lymphatic system include Onchocerca volvulus (river blindness) and Schistosoma haematobium (bilharzia).
CLASSIFICATION OF LYMPHATIC SYSTEM DYSFUNCTION
Whether it is congenital or acquired, lymphatic system dysfunction can be classified according to the type of dysplasia present as follows: lymphangiodysplasia— dysplasia of the lymphatics (LAD I); lymphadenodysplasia—lymph node dysplasia (LAD II); or lymphangioadenodysplasia—dysplasia of the lymphatics and lymph nodes (LAAD).10-12 The classification is similar to that of the International Society for the Study of Vascular Anomalies (ISSVA), which classifies the congenital lymphatic dysplasias as truncal or extratruncal, depending on the embryonic stage at which the defect was produced.13 LAD I dysplasias include agenesis, hypoplasia, disvalvulation, avalvulation, aganglionosis (angioneurosis), lymphangiectasia, lymphangiomatosis, and lymphangiomiomatosis. LAD II dysplasias include hypoplasia, hyperplasia, agenesia, peripheral, central, and global fibrosis, lymphangiomatosis, and intranodal vascular hamartomatosis. Mixed or combined vascular/lymphatic anomalies and malformation syndromes and conditions include the Turner, Noonan, Klippel-Trenaunay-Weber, and Klippel-Trenaunay- Servelle syndromes, Gorham Stout Haferkamp Syndrome, Proteos Syndrome and others.
Using the above classification, a total of 21 causes of dysplasia can be identified. These form the basis of nearly 100 syndromes that are recorded in detail by the Online Mendelian Inheritance in Man (OMIM), the London Dysmorphology Database, the Human Cytogenetic Database, and others.14 Genetic defects are associated with three well-known syndromes.15 Milroy’s disease is a congenital form of primary lymphedema with autosomal dominant inheritance accounting for 2% of primary lymphedema cases. The syndrome is associated with dysfunctional initial lymphatics (hypoplasia of the capillaries) and a failure of absorption of interstitial fluid. Mutations in the VEGFR-3 gene on chromosome 5 are responsible.16 Lymphedema-distichiasis syndrome is associated with lymphatic valve failure giving rise to lymph reflux. This disorder is characterized by distichiasis (a double row of eyelashes) with facial edema at birth and bilateral lower limb lymphedema at puberty. Other features include congenital heart defects and varicose veins.17 This autosomal dominant condition is attributable to a mutation in FOXC2.18 Finally, mutations in the transcription factor SOX18 have been identified in recessive and dominant forms of hypotrichosis–lymphedema–telangiectasia syndrome.19 Lymphedema may be present at birth, or may not be evident during the early years of life, despite the presence of vessel impairment.12 The severity of the lymphedema may be classified according to the clinical features and degree of limb volume from subclinical lymphedema (Grade 0) to fibrotic tissue with skin changes (Grade III).20 Primary lymphedema Grade 0 is not generally diagnosed in pediatric patients. Regardless of the type of primary lymphedema, the underlying causes (LAD I, LAD II, LAAD) are likely to be the same, which has important implications for treatment, as the sooner treatment is initiated the better the possible outcome is likely to be.
Primary lymphedema encompasses a diverse spectrum of human disease. In addition to the LAD I, LAD II, and LAAD dysplasias, it is associated with endothelial dysfunction, and at least 22 combined syndromes, including the combined angiodysplastic syndromes, linking it to at least 120 different diseases. Less wellknown examples amongst these include aortic coarctation, cleft palate, lissencephaly, yellow nail syndrome,21 distichiasis,22 and Turner or Noonan syndrome. The latter is associated with an unknown percentage of primary lymphedema, lymphangiectasias, and external fistulas of the lymph vessels.23 Many of these syndromes are not easily recognized as they are infrequently encountered and can be confused with other disorders. The diagnosis of lymphedema therefore requires careful attention to patient risk factors and specific findings on physical examination. Adequate evaluation of primary lymphedema is required to determine the underlying cause, eg, too few initial lymphatics, collectors too few or too small, collectors too many or too large, too few lymph nodes or malformed lymph nodes, problem with abdominal lymphatics or thoracic duct. All patients with primary lymphedema should undergo genetic analysis to register and gather data on families with lymphedema.24,25 DNA technology allows researchers to perform linkage analysis. By comparing the DNA samples of members with and without lymphedema, they are able to trace which chromosome is involved, and the location of the specific gene on the chromosome, which has important implications for future research.
HISTOPATHOLOGICAL FINDINGS
As the lymphatic system is distributed throughout the human body, lymphatic system dysfunction is implicated in a wide variety of syndromes and diseases both local and systemic.
Lymphangiectasia
Lymphangiectasia is a condition in which the lymphatics are dilated and is frequently associated with lymphedema. Dilated lymphatics are under increased pressure and leak lymph into the surrounding tissue spaces probably through small fistulas. However, not all lymphatic fistulas are due to lymphangiectasias. Cysts that result from lymphatic capillary dilatation (lymphangiomatosis) can also be a cause and may or may not be associated with edema. Pathologically lymphangiomatosis shows multifocal lymphatic dilations, but does not show the localized proliferation of anastomosing lymphatic channels.
Lymphangioma
A lymphangioma is an abnormal collection of dilated lymphatics that are isolated from the normal lymphatic system. Congenital forms are thought to occur because the embryonic lymph sacs have not connected correctly with the lymphatic system and are not associated with lymphedema. When acquired, lymphangiomas arise from endothelialization of trauma-induced lymphoceles and may be associated with lymphedema. A variety of lymphangiomas exist, including uni- or multilocular, and macro- or microcystic, based on the size of the lymphatic spaces within the malformation (Figure 5, Figure 6). They are usually classified into two major groups based on the depth and size of the abnormal lymph vessels. The superficial vesicles are called lymphangioma circumscriptum. The more deep-seated group includes cavernous lymphangioma (cystic hygroma).26 Lymphangioma can occur in any region of the body in which there is lymphatic drainage. The single most common site of cystic occurrence is in the neck (Figure 7). An uncommon, but possible, site is the muscles and bones (eg, phantom bone disease or bone disappearing syndrome—Gorham-Stout syndrome). Combined hemolymphangiomas may also occur, as may combined vascular syndromes, eg, with hemangiomas and risk of malignization—Gorham-Stout-Haferkamp syndrome. Lymphangiomas are a form of angiodysplasia; they have a normal endothelium and therefore cannot be classified as tumoral. Histology of these lesions demonstrates no proliferative component.
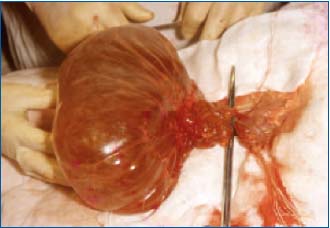
Figure 5. Mesenteric macrocystic, multicystic lymphangioma with
chylous vessels.
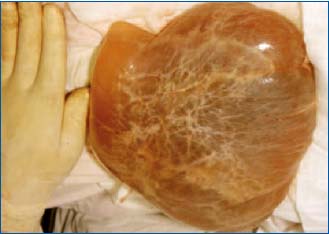
Figure 6. Macrocystic mesenteric lymphangioma.
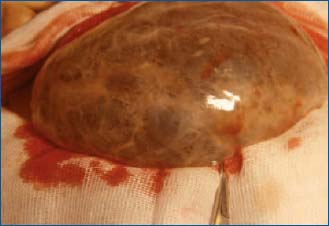
Figure 7. Lymphangiomatosis in a volvulated greater omentum.
Lymphangiomatosis
Lymphangiomatosis is a condition where a lymphangioma is not present in a single localized mass, but in a widespread or multifocal manner. It is often wrongly diagnosed as lymphangioma circumscriptum, but this term describes lymphangioma of thin-walled capillaries. Lymphangiomatosis is frequently associated with other lymphatic-related abnormalities and usually involves multiple organs. The histology of lymphangiomatosis resembles a lymphangioma (Figure 8, Figure 9), but can appear to infiltrate tissues, and may be confused with more aggressive lesions. Intramuscular lymphangiomatosis has not been described, but at this level, phlebo-angiomatosis forms are frequent.27,28
Lymph reflux
Another manifestation of lymphatic dysfunction is systemic lymph reflux, which occurs in both primary and secondary lymphatic dysfunction. The backflow of chyle from the intestines can occur in many areas of the body as a result of abnormalities of the abdominal lymphatics or the thoracic duct. When it leaks into the gut it may lead to lymphedema of the intestinal wall and a generalized swelling of the body as well as protein-losing enteropathy.
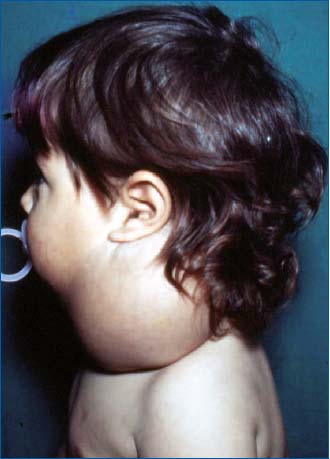
Figure 8. Latero-cervical, macrocystic, unilocular lymphangioma
in a pediatric patient.
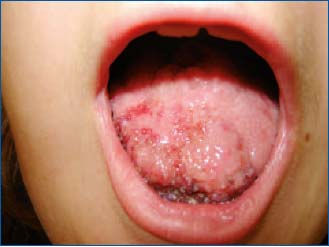
Figure 9. Lymphangiomatosis on a tongue in a pediatric patient.
AREAS OF ONGOING RESEARCH
Angiodysplastic syndromes
Disorders associated with the transport capacity of the lymphatic system (true lymphedema) such as lymphangiomas, lymphangiectasis, and lymph – angiomatosis are well described. However, lymphatic dysfunction can also occur because of an overload of the lymphatics, eg, as a result of infection or cardiac failure, and sometimes a combination of both types of dysfunction can be observed. More research is needed into lymphatic malformations that have mixed vascular system involvement comprising lymphatic and venous and/or arterial vessels in association with chronic venous hypertension and insufficiency.29 Examples of conditions in which lymphedema is a prominent feature include the osteohypertrophic syndromes (hemangiomatosis and varicose veins), Klippel-Trenaunay-Weber syndrome (precapillary arteriovenous shunts and secondary venous hypertension),30 Klippel-Trenaunay-Servelle syndrome (primary deep venous dysplasia and venous hypertension (Figure 10, Figure 11),30 F.P. Weber syndrome (macro-arteriovenous shunts and secondary venous hypertension), cirsoid aneurysm (multiple macro-arteriovenous shunts and hemangioma), Proteus syndrome,31 and other syndromes with primary deep venous dysplasias. In all the above hypertension of the regional lymph system is probably secondary.
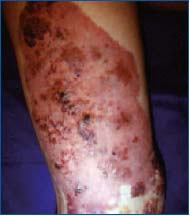
Figure 10. Skin with
hemangioma,
phlebectasias, and
lymphangiectasia in a
patient with Klippel-
Trenaunay-Servelle
syndrome.
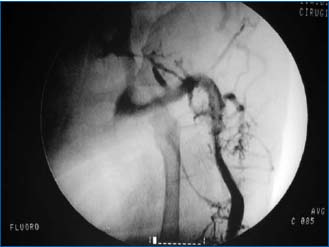
Figure 11. Phlebography findings from a patient with Klippel-
Trenaunay-Servelle syndrome illustrating persistent vena
marginalis (embryonic saphena externa, Ayas or Albanese vein)
confluent to the common femoral vein, with severe hypoplasia of
the superficial femoral vein.
Secondary lymphatic organ dysfunction
Further work is also required on dysfunctions related to the secondary lymphatic organs in relation to immunocompetence. Such research requires a multidisciplinary approach and must be analyzed in the context of multisystem functions in which the lymphatic system is a key player.
CONCLUSIONS
In the past the lymphatic system has played a minor role in traditional pediatric medicine. Although rare, it is now recognized that primary lymphedema is the result of a spectrum of lymphatic disorders. It may be uncomplicated or complicated by other associated disorders of the vascular system. However, with a multidisciplinary team approach, most of these dysfunctions can be diagnosed clinically.

REFERENCES
1. Foldi M, Foldi E. Structural elements of the lymphatic system. In: Foldi E, Foldi M, eds. Foldi’s Textbook of Lymphology. 2nd ed. Munich: Elsevier; 2006:5-13.
2. Castenholz A. Structure of initial and collecting lymphatic vessels. In: Olszewski WL. Lymph Stasis: Pathophysiology, Diagnosis and Treatment. London: CRC Press; 1991:16-39.
3. Baluk P, Fuxe J, Hashizume H, et al. Functionally specialized junctions between endothelial cells of lymphatic vessels. J Exp Med. 2007;204:2349-2362.
4. Zuther JE. Lymphedema Management: The Comprehensive Guide for Practitioners. 2nd ed. New York, NY: Thieme; 2009.
5. Kawamura K, Chung KC. Constriction band syndrome. Hand Clin. 2009;25:257- 264.
6. Papendieck CM. Hair tourniquet syndrome: an unusual cause of perinatologic secondary lymphedema. Lymphology 1999;32:171-172.
7. Price EW. Podoconiosis: non-filarial elephantiasis. 1st ed. Oxford: Oxford Medical Publications, 1990.
8. Wynd S, Melrose WD, Durrheim DN, Carron J, Gyapong M. Understanding the community impact of lymphatic Filariasis: a review of the sociocultural literature. Bull World Health Organ. 2007;85:493-498.
9. Melrose WD. Lymphatic Filariasis: new insights into an old disease. Int J Parasitol. 2002;32:947-960.
10. Barbosa ML, Papendieck CM. Linfangioadenodisplasias en pediatria. Patología Vascular 2000;4:323-327.
11. Papendieck CM. Lymphatic dysplasia in paediatrics. A new classification. Int Angiol. 1999;18:6-9.
12. Papendieck CM. Linfedema en pediatria. Clasificacion y Etiopatogenia. Rev Hosp Niños Buenos Aires. 2005;45:14- 22.
13. Belov S. Geschichte, Epidemiologie und Klassifikation Angeborener Gefäßfehler. In: Loose DA, Weber J, eds. Angeborene Gefäßmißbildungen. Nordlanddruck, Lüneburg, 1997:17-24.
14. Hennekam RC. Syndromic Lymphatic Maldevelopment. In: Witte M, ed. Conquering Lymphatic Disease—NIH Think Tank Conference; May 11–13, 2000; Bethesda.
15. Lymphoedema Framework. Best Practice for the Management of Lymphoedema. International consensus. London: MEP Ltd, 2006.
16. Butler MG, Dagenais SL, Rockson SG, Glover TW. A novel VEGFR3 mutation causes Milroy disease. Am J Med Genet A. 2007;143A:1212-1217.
17. Brice G, Mansour S, Bell R, et al. Analysis of the phenotypic abnormalities in lymphoedema distichiasis syndrome in 74 patients with FOXC-2 mutations or linkage to 16q24. J Med Genet. 2002;39:478-483.
18. Fang J, Dagenais SL, Erikson RP, et al. Mutations in FOXC-2 (MFH-1), a forkhead family transcription factor, are responsible for the hereditary lymphedema-distichiasis syndrome. Am J Hum Genet. 2000;67:1382-1388.
19. Irrthum A, Devriendt K, Chitayat D, et al. Mutations in the transcription factor gene SOX18 underlie recessive and dominant forms of hypotrichosislymphedema- telangiectasia. Am J Hum Genet. 2003;72:470-1478.
20. International Society of Lymphology (ISL). The diagnosis and treatment of peripheral lymphedema. Consensus Document of the International Society of Lymphology. Lymphology 2003;36:84- 91.
21. Nordkild P. Yellow Nail syndrome. Acta Med Scand. 1986;219:221-227.
22. Temple K. Distichiasis-Lymphedema syndrome. Clin Dysmorphol. 1994; 3:139-142.
23. Papendieck CM, Flores M, Gruñeiro L, Bergada C. Síndrome de Noonan con linfangiectasia generalizada. Rev Hosp Niños Buenos Aires. 1974;62.
24. Brouillard P, Vikkula M. Genetic causes of vascular malformations. Hum Mol Genet. 2007;16 Spec No.2:R140-149.
25. Witte MH, Bernas MJ, Martin CP, Witte CL. Lymphangiogenesis and lymphangiodysplasia: from molecular to clinical lymphology. Microsc Res Tech. 2001;55:22-145.
26. Mulliken JB, Young AE. Vascular birthmarks: hemangiomas and malformations. Philadelphia: WB Saunders, 1988.
27. Papendieck CM, Barbosa ML, Pozo PD. Angiodisplasias y grandes síndromes angiodisplásicos en pediatría. In: Simkin R (ed), Tratado de Patología Venosa y Linfatica. Buenos Aires: Medrano Ediciones, 2008;41:851-875.
28. Papendieck CM, Barbosa L, Pozo P. Angiodisplasias em pediatría. Linfedema primario angiodisplasico. Classificação. In: Thomaz JB, Belczak CEQ (eds), Tratado de flebologia e linfologia. Rio de Janeiro: Livraría e Editora Rubio Ltda, 2006;65:767-785.
29. Bergan J. Lymphatic dysfunction in chronic venous insufficiency. 4th National Lymphedema Network International Conference. Orlando 2000. Abstracts p 30.
30. Papendieck CM, Barbosa ML, Pozo P, Braun D, Vannelli C. Klippel- Trenaunay-Servelle syndrome in pediatrics. Lymphat Res Biol. 2003;1:81- 84.
31. Wiedemann HR, Burgio GR, Aldenhoff P, Kunze J, Kaufmann HJ, Schirg E. The proteus syndrome. Partial gigantism of the hands and/or feet, nevi, hemihypertrophy, subcutaneous tumors, macrocephaly or other skull anomalies and possible accelerated growth and visceral affections. Eur J Pediatr. 1983;140:5-12.
