Progress in the management of early thrombus removal
Efthymios Avgerinos MD, FACS, FEBVS
Department of Vascular Surgery,
Attikon Hospital, University of
Athens, Greece
Clinic of Vascular and Endovascular
Surgery, Athens Medical Center,
Greece
Houman Jalaie, , MD, PhD
European Venous Center,
Department of Vascular Surgery,
University Hospital RWTH Aachen,
Germany
ABSTRACT
Deep venous thrombosis (DVT) is common and can be a source of morbidity by way of short-term disabling symptomatology and mid-long term postthrombotic syndrome (PTS). Randomized trials and prospective studies have demonstrated both early and late symptomatic benefit in early recanalization of the iliocaval system of selected patients. On the basis of emerging evidence, published guidelines recommend early thrombus removal in iliofemoral DVT in symptomatic good-risk patients. In light of these recommendations, catheter-directed thrombolysis (CDT) and/or mechanical thrombectomy (MT) have become more popular among vein specialists. This review article summarizes current evidence, novel technologies, and the technical approach to the management of iliofemoral DVT.
Introduction
Over the past 15 years, interventional therapies targeting early thrombus removal have evolved as a potentially better alternative for good-risk patients with iliofemoral deep venous thrombosis (DVT), aiming to decrease postthrombotic syndrome (PTS) rates or severity and improve quality of life (QOL). DVT intervention has steadily migrated toward more minimally invasive techniques in the form of catheter-directed thrombolysis (CDT) and mechanical thrombectomy (MT). Whereas indications and appropriate patient selection are still an area of controversy, increasing awareness, mounting evidence, and experience have earned these modalities a favored place in iliofemoral DVT management.
What have we learned from the lytic trials?
CDT—from early observational cohort studies, comparative nonrandomized, and small randomized studies—appeared to be associated with increased vein patency, valve preservation, and a reduction in the incidence of PTS compared with conventional anticoagulation therapy alone.1-4
The first large, randomized trial was the Norwegian CaVenT trial (Catheter-Directed Venous Thrombolysis in Acute Iliofemoral Vein Thrombosis). The investigators recruited 209 patients, half of them with an iliofemoral DVT.5 Patients were randomized to standard anticoagulation alone or CDT plus anticoagulation. At 24 months, PTS (Villalta score ≥5) developed in 41% of patients in the CDT group and 56% of patients in the standard anticoagulation therapy group (P=0.047). Of note, major bleeding events during the index hospitalization occurred in 2.9% of patients. No bleeding events occurred in the anticoagulation-alone group. At 5 years, the rates of PTS were 43% in the CDT group and 71% (P< 0.0001) in the control group. No difference was found in QOL.6
The ATTRACT trial (Acute venous Thrombosis: Thrombus Removal with Adjunctive Catheter-directed Thrombolysis), the largest randomized controlled trial (RCT) to date involved 691 patients with iliofemoral or femoropopliteal DVT.7 They were randomized to standard anticoagulation therapy alone or pharmacomechanical thrombolysis (PMT) plus anticoagulation. PMT involved predominately the use of the AngioJet device (Boston Scientific Corporation, Marlborough, Massachusetts, USA). At 24 months, PTS (Villalta score ≥5) was 47% in the CDT group and 48% in the anticoagulation-alone (control) group (P=0.56) indicating no benefit for an intervention. Again, bleeding events were more frequent in the procedural group (1.7% vs 0.3%) although none was cerebral or life threatening. A subgroup analysis of the 311 patients with iliofemoral DVT demonstrated that moderate or severe PTS (Villalta score ≥10) was present in 18% in the CDT group and 28% in the anticoagulation group (P=0.021) and severe PTS (Villalta score ≥15) was present in 8.7% in the PMT group and 15% in the anticoagulation group (P=0.048).8 At 30 days, the mean reduction in pain score from baseline was -2.36 in the PMT group and -1.80 in the anticoagulation group (P=0.0082). Mean QOL score at 24 months was 21.5 in the PMT group and 16.2 in the anticoagulation group (P=0.043). Although the primary end point in the ATTRACT trial was not reached, in patients with iliofemoral DVT, PMT resulted in reduction in PTS of any severity using the venous clinical severity score (VCSS), reduction in moderate or severe PTS, reduction in pain and swelling, and improved disease-specific QOL.8-10
The most recent randomized study is the Dutch CAVA trial (CAtheter Versus Anticoagulation alone for acute primary [ilio] femoral DVT), which compared ultrasound-accelerated CDT (EKOS, Boston Scientific Corporation) against anticoagulation for acute iliofemoral DVT.11 This trial recruited 162 patients. Major bleeding occurred in 5% of patients in the CDT group, and no bleeding in the control group. At 12 months, there was no statistical difference in PTS (Villalta score ≥5) between groups: it occurred in 29% of patients in the interventional group and 35% in the anticoagulation alone group (P=0.42). However, a difference in PTS incidence was shown after a median follow-up of 39 months, with reported rates being 47% in the intervention group versus 69% in the group with standard therapy (P=0.01). This difference was the result of a significantly higher number of new diagnoses of mild PTS at the final follow-up visit in the anticoagulation group. For neither definition of PTS was a clinically meaningful change in any of the patient-reported QOL scores demonstrated.12
The conflicting results of the existing RCTs have raised criticism mainly toward diverse patient inclusion criteria or technical variations (eg, stenting rates, timing of intervention, inflow optimization, etc).9,10,13 However, there is little doubt that these trials demonstrated that a certain population can benefit in terms of PTS severity reduction and QOL improvement, and this benefit extends beyond 2 years after treatment.9,10-14 The lytic trials confirmed this benefit in good-risk symptomatic patients with iliofemoral DVT, provided intervention is done early enough, ideally within a 2-week window.13,14 Based on these data, the European Society of Vascular Surgery in its most recent guidelines is recommending early thrombus removal strategies in selected symptomatic patients with iliofemoral DVT (Level of evidence A, recommendation class IIa [evidence in favor of efficacy]).15
We need to acknowledge though that CDT has been associated with higher rates of blood transfusion, pulmonary embolism (PE), bleeding events, and vena cava filter placement. In some countries, CDT is also associated with longer hospital stays and 3 times the hospital costs.16 These are conclusions that have all derived from the lytic trials; however, contemporary practice is shifting away from pure CDT techniques and, without abandoning it, MT is inevitably rising.
Thrombolytics delivered through standard multi-sidehole catheters or in the form of ultrasound-accelerated thrombolysis (EKOS System), for the patient who has minimal bleeding risk, still remain relevant and essential in certain cases as follows: i) to establish inflow in an “ascending” thrombosis (eg, patient with iliofemoral and tibial/popliteal DVT); ii) in any patient whose collateral iliocaval flow is thrombosed and vital to be established (eg, inferior vena cava aplasia); iii) in-stent thrombosis; iv) in inferior vena cava (IVC) filter thrombosis; or v) in extensive bilateral DVT to debulk the large amount of fresh thrombus before initiating MT. It should also be noted that thrombolytics can always bail out an unsuccessful MT procedure as well as an MT procedure can bail out an unsuccessful CDT.
Shifting toward mechanical thrombectomy
Over the past 5 years, contemporary thrombectomy techniques have evolved toward thrombolytic free interventions, altering the safety profile and the complex hospital logistics (eg, need for intensive care unit [ICU] stay). Current practice has shifted toward MT, with a single-session treatment with no ICU stay. Multiple thrombectomy devices are available in the market, but an individual analysis of each one of them is beyond the scope of this document (Figure 1).17-21

Figure 1. Contemporary mechanical thrombectomy devices: AngioJet (Boston Scientific Corporation), ClotTriever (Inari Medical), JETi (Abbott), Aspirex (BD Medical), Indigo CAT 12 (Penumbra), AngioVac (AngioDynamics).
There is mounting evidence from ongoing registries and institutional series on their safety and effectiveness, but there is no long-term (>2 years) data on PTS prevention and no comparative analysis against anticoagulation. Of note, the first RCT was recently initiated, and it is industry sponsored by Inari Medical (DEFIANCE: RCT of ClotTriever System Versus Anticoagulation in Deep Vein Thrombosis).22 This RCT will enroll 300 patients from up to 60 centers worldwide to compare MT with anticoagulation alone for the treatment of iliofemoral DVT. The primary end point for the trial is a hierarchical composite of treatment failure and PTS syndrome severity at 6 months.
Technical aspects for optimal outcomes
Before the decision to intervene, thrombus within the iliofemoral segment needs to be confirmed. Aside from a baseline duplex scan, a cross sectional imaging MR venogram or computed tomography (CT) venogram (abdomen, pelvis, and upper leg) can facilitate operative planning by documenting the extent of the thrombus and uncovering unusual anatomies (eg, duplicated cava, IVC aplasia, etc) or chronic venous obstruction. Decision-making and technique selection are summarized in Figure 2.
Access
With few exceptions, treating an iliofemoral DVT will require popliteal vein access with the patient in prone positioning. This will guarantee good control and imaging of the femoral bifurcation that is the gatekeeper of iliac vein patency. The presence of popliteal thrombus is not a contraindication to access the vein. Use of ultrasound and a micropuncture system is recommended to minimize bleeding complications, particularly if CDT is considered. If needed, the popliteal vein can accept large sheaths to accommodate the standard venous stent delivery systems (9 or 10 Fr) and even up to 16 Fr after serial dilatations for larger thrombectomy devices. Proximal tibial or small saphenous access can also be obtained and can accommodate 9- to 10-Fr sheaths. An ipsilateral mid-femoral puncture in the supine position can also be sufficient for isolated iliac or caval DVT.
A 5-Fr short sheath, a starter 0.035-inch wire, and a standard guiding catheter are typically enough to cross fresh thrombus and obtain images at the femoropopliteal and iliocaval segments.
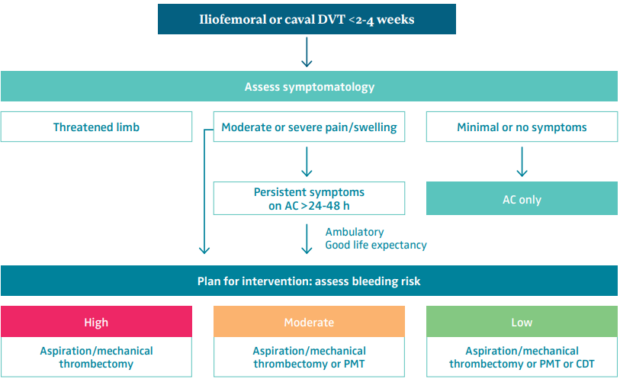
Figure 2. Patient and treatment selection for iliofemoral deep venous thrombosis. Abbreviations: AC, anticoagulation; CDT, catheter-directed thrombolysis; DVT, deep venous thrombosis; PMT, pharmacomechanical thrombectomy.
Thrombolysis, thrombectomy, and IVC filters
Thrombolysis can be performed using a multi-sidehole standard catheter or the EKOS catheter that incorporates ultrasound probes to accelerate fibrin separation. It is essential to accommodate the entire infusion catheter segment (available is several lengths 5-50 cm) within the clot; otherwise, the lytic agent will escape through the holes of least resistance into the blood stream and not within the clot. Contemporary thrombolysis protocols can vary in time between 6 to 12 hours, and dosage should typically range between 0.5 to 2 mg/hour. Patients will need to be transferred to the intensive care unit for monitoring during the dripping and returned to the interventional suite for termination of the procedure. Frequently, extension of CDT up to 48 hours or additional MT may be required to maximize clot removal.
Aspiration or MT can be performed with any of the novel available devices on the market (Figure 1) with which the practitioner feels comfortable. A 10- to 16-Fr sheath will typically be required. The ultimate target of a successful thrombus removal is >90% extraction and provides optimal inflow (through the femoral and deep femoral veins) to the iliac segment. This will minimize the risk of early re-thrombosis or later PTS.23-26
Regarding the use of IVC filters, whereas a small, randomized trial has indicated a higher rate of clinically significant PE in patients not receiving one, there was no mortality difference, and subsequent contemporary studies recommend highly selective IVC filtration.27 PE can be unavoidable, but they are rarely clinically meaningful for otherwise good-risk patients, and placement of an IVC filter may introduce complexity and other potential risks. Patients that might benefit are those with associated PE on presentation, with large mobile thrombus, or those who are planned for aggressive pharmacomechanical thrombectomy involving the IVC.28
Intravascular ultrasound
Intravascular ultrasound (IVUS) allows for detailed images acquired in an axial plane relative to the catheter tip; it reduces radiation exposure and contrast volume in the typically young patient. IVUS has been shown in multiple studies to be superior for accurate lesion identification compared with plain venography, and its use improves long term patency.29 In contemporary acute DVT intervention, IVUS is essential not only to identify residual clot and external compression but also to guide choice of stent diameter and landing zones, and to confirm a satisfactory final outcome (eg, stent expansion).
Additionally, distance markers on the catheter shaft can be used for precise length measurements. If IVUS is not available, multiple venographic projections should be obtained for lesion identification.
Venous stents
Several dedicated venous stents are available (Figure 3). Sufficient stenting of persistent lesions (chronic obstruction, residual thrombus, external compression) following thrombus removal seems to be a critical component of a clinically successful procedure (Figure 4). Accumulated experience favors liberal stenting and use of dedicated venous stents to ensure good inflow and outflow. Consequently, in many cases it may be necessary to stent from the iliocaval confluence down to the common femoral vein.25 Care should be taken to prevent jailing of contralateral common iliac vein as well as jailing of the deep femoral vein when extending distally. The common iliac vein is typically stented with a 14- to 16-mm stent, and the external iliac/common femoral veins with a 12- to 14-mm stent. The length of the iliac stent should also be long enough (≥8 cm) to anchor at the external iliac segment, preventing migration and avoiding an acute angulated landing at the iliosacral curvature.
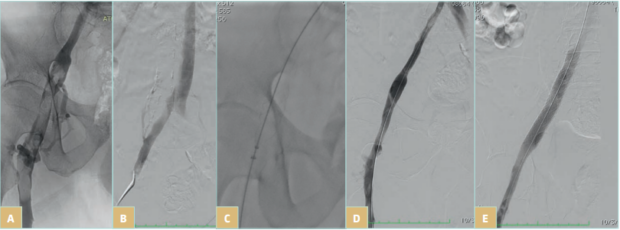
Figure 4. Symptomatic iliofemoral deep venous thrombosis (DVT) managed with ClotTriever (Inari Medical) mechanical thrombectomy and placement of a 14x 100 m BeYond (Bentley) stent: A) thrombus at the iliofemoral segment extending distally to the origins of the femoral and deep femoral veins; B) clean but diseased proximal iliac segment (notice the presence of collateral vein); C) mechanical thrombectomy (notice the coring element and the collection bag of the ClotTriever device); D) venogram post thrombectomy with almost complete thrombus resolution but residual stenosis (confirmed also with intravascular ultrasound); and E) final venogram after stent placement indicating brisk flow through the previously thrombosed iliofemoral segment.
Perioperative care and surveillance
The patient should remain on bed rest for 2 to 4 hours to allow for hemostasis; the index leg needs to be tightly wrapped, and after hospital discharge, thigh-high compression at 20 to 30 mm Hg should be encouraged for at least 1 month or until the swelling completely resolves. The patient should also be encouraged to drink plenty of fluids in order to minimize the effects of hemoglobinuria. Within 6 to 8 hours, the patient should be encouraged to ambulate.
The patient should be discharged with a defined plan for anticoagulant therapy that is consistent with their risk of recurrence.15,30 For patients who received a stent, before initiating oral anticoagulation, low molecular weight heparin for 2 to 6 weeks is preferred owing to its anti-inflammatory effects. An antiplatelet agent for 6 months or indefinitely depending on the patient’s risk profile can be considered. Appropriate referral to hematology is warranted in patients with an unprovoked DVT or possible thrombophilia. A follow-up office visit is recommended at 2 to 4 weeks, at 3, 6, and12 months, and annually thereafter with duplex ultrasound.15,30,31 Cross-sectional imaging can be needed on occasion in complex iliocaval reconstructions to evaluate patency.
Conclusions
Mounting evidence demonstrates early symptomatic relief and PTS severity reduction with early percutaneous DVT debulking. Although anticoagulation and compression remain the mainstay of treatment, patients with iliofemoral DVT associated with swelling and pain, and good life expectancy, should be strongly considered for treatment with a minimal invasive catheter intervention. Contemporary procedures are generally safe and do not require prolonged hospitalization. As DVT rates are rising, awareness of novel treatments and appropriate technical expertise within a multidisciplinary team can guarantee optimal results and ultimately a better QOL for our patients.
CORRESPONDING AUTHOR
Efthymios Avgerinos, , MD, FACS, FEBVS
Athens Medical Center; Athens, Greece
email: eavgerinos@vascularhealth.gr
References
1. Comerota AJ. Catheter-directed thrombolysis for the treatment of acute iliofemoral deep venous thrombosis. Phlebology. 2001;15:149-155.
2. Markevicius N, Apanavicius G, Skerbinskas S. Comparison between long term results of catheter directed thrombolysis and anticoagulation in the treatment of acute iliofemoral deep vein thrombosis. Phlebology. 2004;19:148-149.
3. Broholm R, Sillesen H, Damsgaard MT, et al. Postthrombotic syndrome and quality of life in patients with iliofemoral venous thrombosis treated with catheter-directed thrombolysis. J Vasc Surg. 2011;54(6 Suppl):18S-25S.
4. Elsharawy M, Elzayat E. Early results of thrombolysis vs anticoagulation in iliofemoral venous thrombosis. A randomised clinical trial. Eur J Vasc Endovasc Surg. 2002;24:209-214.
5. Enden T, Haig Y, Klow NE, et al. Long-term outcome after additional catheter-directed thrombolysis versus standard treatment for acute iliofemoral deep vein thrombosis (the CaVenT study): a randomised controlled trial. Lancet. 2012;379(9810):31-38.
6. Haig Y, Enden T, Grøtta NEK, et al. Post-thrombotic syndrome after catheter-directed thrombolysis for deep vein thrombosis (CaVenT): 5-year follow-up results of an open-label randomised controlled trial. Lancet Haematol. 2016;3:e64-e71.
7. Vedantham S, Goldhaber SZ, Julian JA, Kahn SR; for the ATTRACT trial. Pharmacomechanical catheter-directed thrombolysis for deep vein thrombosis. N Engl J Med. 2017;377(23):2240-2252.
8. Comerota AJ, Kearon C, Gu CS, et al. Endovascular thrombus removal for acute iliofemoral deep vein thrombosis: analysis from a stratified multicenter randomised trial. Circulation. 2019;139(9):1162- 1173.
9. Avgerinos EA, Chaer RA. The ATTRACTiveness of catheter-directed thrombolysis. J Vasc Surg Venous Lymphat Disord. 2018;6(3):303.
10. Nana P, Avgerinos E, Spanos K, Giannoukas A, Labropoulos N. Gaps arising from randomized controlled trials on thrombolysis for proximal deep vein thrombosis of the lower limb. J Vasc Surg Venous Lymphat Disord. 2022;10(1):196- 199.
11. Notten P, ten Gate-Hoek AJ, Arnoldussen CWKP, et al. Ultrasound-accelerated catheter-directed thrombolysis versus anticoagulation for the prevention of post-thrombotic syndrome (CAVA): a single blind, multicentre, randomised trial. Lancet Haematol. 2020;7(1):e40-e49.
12. Notten P, de Smet AAEA, Tick LW, et al. CAVA (Ultrasound-accelerated catheter-directed thrombolysis on preventing post-thrombotic syndrome) trial: long-term follow-up results. J Am Heart Assoc. 2021;10:e018973.
13. Go C, Chaer RA, Avgerinos ED. Catheter interventions for acute deep venous thrombosis: who, when and how. Vasc Endovasc Rev. 2020;3:e07.
14. Javed A, Machin M, Gwozdz AM, et al. Meta-analysis of lytic catheter-based intervention for acute proximal deep vein thrombosis in the reduction of post thrombotic syndrome. J Vasc Surg Venous Lymph Disord. 2023;11(4):866-875.
15. Kakkos SK, Gohel M, Baekgaard N, et al. European Society for Vascular Surgery (ESVS) 2021 Clinical Practice Guidelines on the Management of Venous Thrombosis. Eur J Vasc Endovasc Surg. 2021;61:9-82.
16. Bashir R, Zack CJ, Zhao H, Comerota AJ, Bove AA. Comparative outcomes of catheter-directed thrombolysis plus anticoagulation vs anticoagulation alone to treat lower-extremity proximal deep vein thrombosis. JAMA Intern Med. 2014;174:1494-1501.
17. Harrison B, Hao F, Koney N, McWilliams J, Moriarty JM. Caval thrombus management: the data, where we are, and how it is done. Tech Vasc Interv Radiol. 2018;21:65-77.
18. Lichtenberg M, Stahlhoff WF, Özkapi A, de Graaf R, Breuckmann F. Safety, procedural success and outcome of the Aspirex S endovascular thrombectomy system in the treatment of iliofemoral deep vein thrombosis – data from the Arnsberg Aspirex registry. Vasa. 2019;48:341-346.
19. Lopez R, DeMartino R, Fleming M, Bjarnason H, Neisen M. Aspiration thrombectomy for acute iliofemoral or central deep venous thrombosis. J Vasc Surg Venous Lymphat Disord. 2019;7:162-168.
20. Razavi C, Khalsa B, Openshaw L, Razavi MK. Single-session treatment of patients with symptomatic iliocaval and iliofemoral deep vein thrombosis: technical results of a prospective pilot study. J Vasc Interv Radiol. 2022;33:183-188.
21. Dexter DJ, Kado H, Schor J, et al. Interim outcomes of mechanical thrombectomy for deep vein thrombosis from the All-comer CLOUT Registry. J Vasc Surg Venous Lymphat Disord. 2022;10:832-840.e2.
22. Inari Medical begins DEFIANCE randomized clinical trial of ClotTriever system in DVT. Published January 12, 2023. Endovascular Today. https://evtoday.com/news/inari-medical-begins-defiance-randomized-clinical-trial-of-clottriever-system-in-dvt
23. Aziz F, Comerota AJ. Quantity of residual thrombus after successful catheter-directed thrombolysis for iliofemoral deep venous thrombosis correlates with recurrence. Eur J Vasc Endovasc Surg. 2012;44:210-213.
24. Comerota AJ, Grewal N, Martinez JT, et al. Postthrombotic morbidity correlates with residual thrombus following catheter-directed thrombolysis for iliofemoral deep vein thrombosis. J Vasc Surg. 2012;55:768-773.
25. Avgerinos E, Saadeddin Z, Abou Ali AN, et al. Outcomes and predictors of failure of iliac vein stenting after catheter-directed thrombolysis for acute iliofemoral thrombosis. J Vasc Surg Venous Lymphat Disord. 2019;7:153-161.
26. Pouncey AL, Kahn T, Morris RI, Saha P, Thulasidasan N, Black SA. Risk factors and classification of reintervention following deep venous stenting for acute iliofemoral deep vein thrombosis. J Vasc Surg Venous Lymphat Disord. 2022;10:1051-1058.
27. Sharifi M, Bay C, Skrocki L. Role of IVC filters in endovenous therapy for deep venous thrombosis: The FILTER-PEVI (filter mplantation to lower thromboembolic risk in percutaneous endovenous intervention) trial. Cardiovasc Intervent Radiol. 2012;35:1408-1413.
28. Avgerinos ED, Hager ES, Jeyabaln G, Marone L, et al. Inferior vena cava filter placement during thrombolysis for acute iliofemoral deep venous thrombosis. J Vasc Surg Venous Lymphat Disord. 2014;2:274-281.
29. Tran LM, Go C, Zaghloul M, et al. Intravascular ultrasound evaluation during iliofemoral venous stenting is associated with improved midterm patency outcomes. J Vasc Surg Venous Lymphat Disord. 2022;10:1294-1303.
30. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST Guideline and Expert Panel Report. CHEST. 2016;149:315-352.
31. Abdul-Haqq R, Pearce BJ, Matthews TC, Paterson MA, Jordan WD Jr, Passman MA. Routine extended follow-up surveillance of iliac vein stents for iliocaval venous obstruction may not be warranted. J Vasc Surg Venous Lymphat Disord. 2017;5:500- 505.

