Radiofrequency ablation of the incompetent saphenous vein-lessons learned
INTRODUCTION
Investigations into the therapeutic use of radiofrequency (RF) energy in man occurred as early as the late 19th and early 20th centuries. Technological advances increased interest in RF applications. Because of its precise control of energy delivery and reliability, RF energy has been used for decades in neurosurgical techniques.1 By the 1980s cardiac arrhythmias were being treated with RF devices.2 Usage expanded to include treatment of various malignancies (including hepatic, renal, musculoskeletal, breast, lymph, spleen, pulmonary),3,4 as well as ophthalmologic maladies, gastric reflux, sleep apnea, and aesthetic dermatological conditions.5,6 Berjano reported that the number of scientific papers published on the topic of therapeutic RF energy use increased from 19 in 1990 to 825 in 2005.5 As a less invasive alternative to vein stripping for elimination of saphenous vein reflux, the percutaneous catheter-based radiofrequency Closure® procedure (VNUS Medical Technologies, San Jose, CA) was introduced in Europe in 1998 and in the U.S. in 1999.
Following initial experience with the Closure® procedure and early technique modifications, it became clear that reflux at the saphenofemoral junction (SFJ) could be eliminated by obliteration of the great saphenous vein in the thigh without resorting to dissection and ligation of all contributing branches near the saphenofemoral junction,7,8 thus eliminating the need for a groin incision and potential for minor and even major complications that can occur following traditional ligation and stripping procedures, and leaving intact venous return and lymphatic drainage from the abdominal wall and lower extremity. The validity of this strategy has been borne out by several published mid-term reports.9-12 Pichot11 coordinated an extensive two-year follow-up ultrasound evaluation study from five VNUS Registry centers. The results showed that 58/63 (92.1%) treated GSV segments remained free of reflux. Junctional tributary reflux was seen in 7/63 (11.1%) limbs, four of which were associated with the SFJ as the sole source of reflux. Neovascularization was not observed in any treated limbs. More recently, Closure® equipment innovations and technique modifications have contributed to reduced procedure times while maintaining efficacy and low rates of complications.
TECHNIQUE
Unlike earlier attempts to obliterate the saphenous vein by diathermy, the endovenous Closure® procedure uses radiofrequency energy to heat the vein wall. The ClosurePLUS™ catheter employs intralumenally positioned bipolar electrodes located at the tip in contact with the vein wall. Electrical current flowing between the electrodes through the vein wall tissue generates heat by a phenomenon called “resistive heating”. The new ClosureFAST™ catheter (illustrated in Figure 1) utilizes radiofrequency energy to heat a 7 cm long element near the tip resulting in direct conduction to the vein wall. For satisfactory transfer of energy, the electrodes or the element must be in direct contact with the vein wall. The heating causes a physical shortening of the vein wall’s collagen fibrils in a mostly uniform manner, primarily in the subendothelial layers.13 A recent report by Schmedt et al corroborates this finding.14 The vein narrows while at the same time denatured blood proteins congeal to obliterate the vein lumen. The entire treated vein is affected by this process much like soft boiling an egg. Over the next several months, usually ten to twelve, and certainly by two years, the vein fibroses and is seen to vanish on duplex ultrasound (DUS) in over 86% of cases.
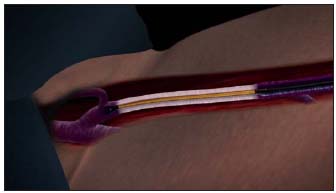
Figure 1. The ClosureFASTTM catheter distal section, which contains a 7 cm long heating element, is depicted heating the GSV vein segment starting approximately 2 cm below the SFJ. (GSV – great saphenous vein; SFJ –saphenofemoral junction)
The ClosurePLUS™ procedure is controlled by a computerized generator (RF1 or RF2) which monitors electrode temperature and adjusts energy levels to achieve a constant heating of the vein wall at a userselectable temperature, typically 85° or 90° ± 3°C. During heating, the catheter is withdrawn from the vein typically at a rate of 2-4 centimeters per minute. For the ClosureFAST™ procedure, the newer RF2 generator (which can be used with all VNUS catheters) controls the heating element at 120°C for a predetermined 20-second period that can be manually interrupted at any moment by the operator. The major steps of the technique as currently practiced at the Reno Vein Clinic are as follows:
Patient anesthesia: Choice of anesthesia is a matter between the physician and the patient. However, the procedure itself is well suited to local anesthesia or regional field block such as a femoral nerve block. General anesthesia has at least one drawback: the inability of the anesthetized patient to communicate nerve pain which might be the result of the heat produced by the catheter coming into proximity with an overlying sensory cutaneous nerve. Minimal sedation with oral (diazepam) or intravenous (midazolam and fentanyl) agents is recommended to provide adequate anxiolysis and analgesia. We have found in our practice that short-acting agents delivered intravenously offer better control, with quick recovery and better patient comfort, thus allowing fast-track postprocedure discharge from the facility or office. Midazolam IV (diazepam family) may offer some protection against lidocaine toxicity.
Catheter insertion: The catheter is inserted into the vein at its point nearest to the skin surface, usually just below or above the knee, using standard percutaneous (Seldinger) or cut-down technique. The catheter tip is positioned using ultrasound guidance approximately 2 cm distal to the saphenofemoral junction. This is critical to avoid thermal injury to the SFJ. All current Closure® devices (VNUS Medical Technologies, San Jose, CA) feature a central lumen that will accommodate either a 0.018 (ClosureRFSTM) or 0.025 inch (ClosurePLUSTM and ClosureFASTTM) diameter guide wire, to allow maneuverability through tortuous or difficult vein segments.
Limb anesthesia and vein compression: Once the catheter has been positioned safely below the SFJ, tumescent anesthesia can be introduced using a variety of methods. It is important to use ultrasound visualization in order to insure that the fluid is placed beneath the saphenous fascia and above the deep muscular fascia and that it surrounds the vein completely, which serves to contain the radiant heat within the treated vein without significantly affecting adjacent cutaneous and sensory nerve tissues.15 The infusate also compresses the saphenous vein and its inflow branches in order to produce a “dry” vein. Contraction of the vein diameter is another benefit if diluted epinephrine is included in the tumescent anesthetic fluid. Tumescent anesthesia, using generous volumes of buffered lidocaine 1% with epinephrine 1:100,000 diluted to 0.1% placed properly, results in relatively pain-free status (Table I). Care must be taken to avoid lidocaine toxicity – dosage guidelines are 7 mg/kg body weight, and no more than 500 mg should be used at one setting. Bilateral limb procedures may require an alternative anesthesia choice, such as general or regional (femoral nerve block, spinal, or epidural).
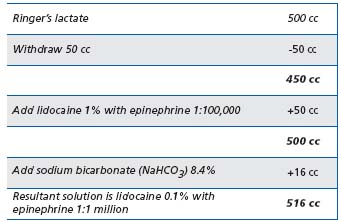
Table I. Tumescent anesthesia solution preparation
Energy delivery: Prior to November 2007, during vein heating the patient was positioned in the Trendelenburg position, with approximately 15-20 degrees of angulation. More recently, as depicted in Figure 2, the patient’s head and torso are kept in a relatively comfortable horizontal position while the limb to be treated is elevated at an angle of 30-45 degrees. Ultrasound imaging demonstrates that, while the patient remains comfortable, this extreme position allows for near complete emptying of the venous system with tight circumferential contact of the vein by the catheter. Gentle manual pressure with the DUS probe can be applied to the saphenofemoral junction area and then along the course of the vein as necessary, while the catheter is withdrawn (ClosurePLUSTM) or segmentally repositioned (ClosureFASTTM).
Perioperative ultrasound: DUS should be used to document satisfactory closure of the treated vein just before removing the catheter from the vein. When ClosurePLUSTM is performed, a curious finding on ultrasound that may be observed is echogenic movement depicted in the occluded vein despite having obliterated the lumen. It probably represents movement of saline solution infused through the Closure® catheter in the vein and around the blood plug despite adequate obliteration of the lumen. If significant flow remains, the vein should be retreated. A word of CAUTION: when using the ClosureFASTTM catheter, NEVER re-advance the catheter to the SFJ, as doing so may dislodge fibrinous material cephalad into the proximal deep venous system. If concerned when treating very large diameter veins, approximately 10 mm or greater, one can simply double heat each 7 cm segment (two episodes, 20 seconds each) prior to repositioning the catheter. The transfer of heat with ClosureFASTTM is so uniform and efficient that the vein treated invariably closes when studied postoperatively with DUS.
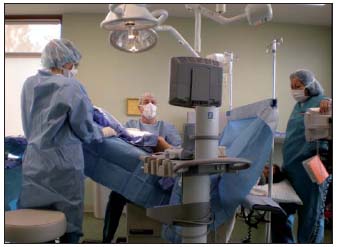
Figure 2. The tilt table in this treatment room allows the torso of the patient to be in a relatively supine position (patient’s head can be seen to the right), while the leg to be treated is positioned with 30° or more of angulation.
Postoperative instructions: The patient is encouraged to ambulate immediately, returning in some cases to normal activities on the same day. Postoperative ultrasound imaging of the saphenofemoral junction within three days is an essential part of the protocol to check for successful obliteration and absence of clot extension into the common femoral vein. What is usually seen at this initial check is remarkably similar to an acute thrombosis of the vein with dilation and filling of the vein lumen with echo dense signals and failure to compress with externally applied pressure. This represents an element of thrombosis which aids the obliteration process
MANAGEMENT OF COMPLICATIONS
Data collected for the VNUS Closure® Study Group Registry was prospective, looking for nerve injury, clot extension, hematoma, phlebitis, skin burns, and infection.16 Only limbs treated at 85ºC and seen within the first postoperative week were included in the report, and only limbs treated with Closure® and ClosurePLUSTM catheters were included in the Registry. Results are shown in Table II. The most serious complication, although rare, is clot extension into the common femoral vein as it can lead to deep vein thrombosis (DVT) if not recognized and treated early with either low-molecularweight heparin (LMWH) or operative thrombectomy. It is the practice at the Reno Vein Clinic to see all cases on the first postoperative day and to include a postoperative duplex scan during that visit. If there is evidence of clot extending into the deep vein, then LMWH is prescribed at therapeutic doses for six days. Aspirin 325 mg is started on day 7 and continued for one month.
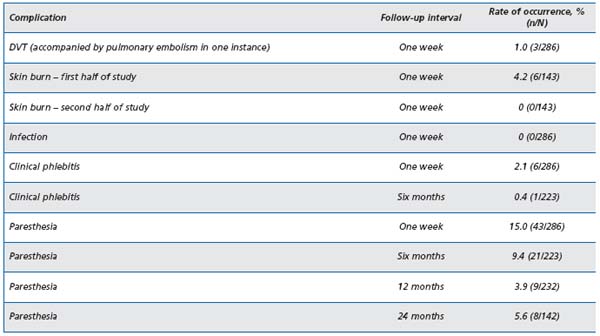
Table II. Complications reported from the Closure Study Group
Nerve injury associated with RF ablation is seen as areas of hypoesthesia noted on follow-up examination in the first week postop. The majority of these occurred in the early Closure® cases before the routine instillation of tumescent anesthesia. To avoid nerve injury, following the early clinical experience, the Closure® procedure was recommended to be limited to above-knee GSV treatments.13 The greater saphenous nerve is actually adherent to the GSV in the distal leg and injury to this nerve is usually unavoidable when GSV RF ablation is attempted much below the knee.8
Skin burns, initially seen in a minority of early Closure® cases, essentially have vanished since the institution of tumescent anesthesia16,17 and the abandonment of the Eschmark leg wrap. The Eschmark rubber bandage has a tendency to roll back when applied to the funnel-shaped thigh, in which case it can act as a tight rubber band to push the skin closer to the saphenous vein. Ablation in the thin or skinny leg should prompt careful attention to detail to minimize thermal injuries to the overlying skin due to excessive external compression, which can arise from the Eschmark bandage or the DUS probe during intraoperative monitoring.
Phlebitis can occur with the Closure® procedure as in any treatment of varicose veins, and it is usually the result of residual blood trapped within vein segments. Some degree of phlebitis is inherent in the whole process since the obliteration occurs as a result of injury to the vein by the heating process. It is occasionally seen as a tender, erythematous or ecchymotic band over the treated vein in the distal thigh. It resolves over several weeks without any specific treatment other than for symptomatic relief, eg, the use of nonsteroidal antiinflammatory drugs, heat, and compression hosiery.
Patients may describe a curious sensation which occurs during the second or third postop week along the treated vein segment, usually in the distal thigh. They may experience a spontaneous or persistent dull feeling, or “bogginess”, or sharpness when stretching or extending the treated leg. This could represent an inflammatory process, which occurs as the body is healing the scald injury of the treated vein segment. The sensations usually abate over several weeks, consistent with the normal healing time of injured tissues.
EXPERIENCES AT THE RENO VEIN CLINIC
At the Reno Vein Clinic our routine practice is to conduct the first postoperative clinical and DUS examination at one day after RF obliteration, with periodic subsequent wellness visits as patient schedules permit. Following procedures performed from 1999 through March 2006 (using ClosurePLUSTM at 90ºC since April 2002), we documented a total of 29 failures from 598 ClosurePLUSTM obliteration procedures having one-day postoperative DUS. All failures were detected at or prior to a three-year follow-up examination. There were 124 duplex examinations conducted beyond three years, 34 of which were five years or more following intervention, with no further failures observed. Only one case of DVT occurred related to aggressive heating near the SFJ. Since instituting routine use of tumescence, only two cases of second degree burns occurred, both on skinny male limbs; and temporary paresthesia rate of only a 2% was seen. Further details on our complications, which parallel the broader experience with the Closure® procedure, were previously reported.18 In three cases in which SFJ reflux abolition was documented as successful one day postoperatively, reflux developed within three months (n=2) and nine months (n=1) through the SFJ and into pre-existing varicose veins via the anterolateral saphenous vein. Because of technical difficulties, significant thigh varicose veins had been left untreated at the time of the Closure® procedure. These three specific personal observations suggest that SFJ incompetence may be secondary to downstream venous insufficiency rather than a primary contributor to superficial venous insufficiency.
Other Saphenous Veins
From January 2002 to March 2006, RF ablation at the Reno Vein Clinic was extended to include several cases of anterior and posterior branches of the GSV in the proximal thigh. These results generally parallel those of the GSV. Also, during that period, 55 small (lesser) saphenous veins (SSVs) were successfully treated, with one instance of temporary sural nerve injury. All SSVs were confirmed closed at the one-day postoperative visit. When 90°C is used, successful short-term and mid-term outcomes appear to mimic those seen in the GSV group. In the case of SSV treatment, careful ultrasound guidance is critical for precise placement of the catheter electrodes to avoid inadvertent heating of the posterior tibial nerve. In our experience, pain located in the heel or foot at onset of heating indicates placement too close to the nerve. Tumescent anesthesia infiltrated circumferentially around the SSV is a must to avoid injury to the sural nerve, which usually lies near the vein.
We have also begun extending the application of temperature-controlled RF vein obliteration to the treatment of incompetent perforators (up to 5 mm diameter) and 3-4 mm diameter short segment refluxing primary or tributary veins such as small saphenous, posterior and anterior saphenous veins. The rigid 2 mmX15 cm long ClosureRFSTM stylet can be introduced over a 0.018” guide wire and positioned under DUS guidance.
CLOSURE® TREATMENT RESULTS
Data on over 1000 limbs treated without high ligation have been collected in an ongoing Registry of the VNUS Closure® Treatment Study Group comprising 35 centers in the US, Europe, and Australia. Early results from this registry at various follow-up periods through January 2002, as reported by Merchant et al, show successful ablation ranging from 93% at one week to 85% at two years, with absence of vein reflux (defined as absence of reversed flow at or near the SFJ or in any segment of the treated vein) of 90% at two years, and patient satisfaction of 95% at two-year follow-up. In addition, 111 of 142 limbs with 2-year DUS examinations were also scanned at 1 year; of these, only two (1.8%) changed from reflux-free at 1 year to DUS evidence of reflux at 2 years.16
In 2005, Merchant et al reported the 5-year Closure Study Group outcomes on 1222 vein segments treated at 85ºC at 34 international centers, and included procedures performed during initial learning curve periods.19 Twelve of the 34 centers provided long-term follow-up. Reflux-free and vein occlusion rates in 117 limbs examined at five years were 83.8% and 87.2%, respectively. Clinical symptom improvement, measured by absence of limb pain, fatigue, or edema, was observed in 85% to 94% of limbs classified as having anatomic success at annual intervals over the 5-year period. Over the same interval, the 185 (15.1%) limbs presenting by DUS examination as anatomical failures exhibited 70% to 80% clinical improvement.
Several prospective randomized studies on the early results of Closure® treatment without high ligation compared with vein stripping and ligation have been published20-23 and show significant clinical superiority of the Closure® procedure. In a study from one center in Oulu, Finland,20 significant advantages of the Closure® treatment were shown regarding less pain, early return to activities, fewer sick leaves from work, and better quality of life scores. When these findings included time lost from work, the authors found Closure® treatment to be cost effective despite initial high hospital costs. A three-year follow-up report demonstrated a durable ablation in all 15 RF-treated limbs.24
Another study, EVOLVeS, supported by VNUS Medical Technologies, involved five centers in the US and Europe,21 and was designed to determine the early benefits of the procedure with follow-up limited to four months. Three-week results showed significant advantage of the Closure® procedure in that there was less pain, earlier return to activities and work, better quality of life scores, and better cosmetic results. When these patients were seen at the four-month follow-up, these advantages had disappeared. Although the study was not designed to evaluate cost effectiveness, when the severity of infectious complications (which occurred only in the stripping and ligation group) were factored in, the authors ascribed a probable cost benefit to Closure®.
A one- and two-year follow-up of this multicenter study by Lurie et al12 showed that 41% of obliterated GSVs became ultrasonically undetectable, and another 51% remained visible, but exhibited progressive diameter shrinkage. Vein remnants that remained visible by DUS were larger at the time of RF ablation than those that became invisible. Clinical status of limbs that underwent RF ablation was at least equal to the status of limbs that received vein stripping. The cumulative rate of recurrence of varicose veins was 14.3% in the RF group compared with 20.9% in the stripping group. Using the CEAP classification system, 33% of RF patients and 28% of stripping patients had no signs of venous disease at two years. Quality of life questionnaires were administered at all follow-up visits. While the observed superiority of the RF group over the stripping group diminished by four months, it re-emerged at both the one- and two-year intervals.
PERFORATING VEINS
Whiteley et al reported the first application of the ClosurePLUSTM catheter in treating incompetent perforating veins (IPVs) in 2003.25 The procedure was performed on 770 IPVs in 506 limbs. At one year, 79% of IPVs were closed and had atrophied; 76% remained closed at 2 years. Encouraged by this early success, VNUS Medical Technologies designed Radiofrequency Stylet devices and implemented a standard protocol for the ablation of clinically significant IPVs. Peden and Lumsden, generally restricting the application to patients in the CEAP 4-6 categories and emphasizing the need to adequately treat the subfascial component of the IPV, described their technique for the treatment of IPVs and have reported early successes similar to those of Whiteley et al.26
SAPHENOFEMORAL CONFLUENCE
Concern about the fate of the saphenofemoral confluence following elimination of the main trunk of the saphenous vein has been addressed by several reports.
Gradman, in a survey of members of the American Venous Forum and American College of Phlebology, concluded that regardless of the method of saphenous vein ablation (RF, laser, or foam sclerotherapy), concomitant ligation of the saphenofemoral junction offered no advantage in outcome no matter the size of the proximal great saphenous vein.27 Boros et al, in a single community center retrospective analysis of outcomes of saphenous vein RF ablation, found no difference whether the junction was routinely, selectively, or never ligated: the DVT rate of 4% was equal across the board. The infection rate for SFJ ligation (5%) was limited to the groin incision and was significantly higher (P=0.01) than when no ligation was performed (0%).28 In a retrospective study analyzing great saphenous vein stripping with subjunctional ligation (thus preserving the saphenofemoral confluence of tributaries), Pittaluga et al observed no reflux in the saphenofemoral confluence in 98.2% of cases with a mean follow-up of 24.4 months.29 This finding is comparable to that of Pichot et al regarding analysis at 2 years following RF ablation without high ligation.11
Regarding clinical usefulness of the RF ablation techniques, Frasier et al found successful treatment of selected Klippel-Trenaunay syndrome patients.30 Vasquez et al reported successful venous stasis ulcer healing and the usefulness of the Venous Clinical Severity Score (VCSS) in assessing outcomes after saphenous vein RF ablation.31 Puggioni et al in a retrospective review of case studies at the Mayo Clinic concluded that postoperative complications, in their experience, were less frequent with RF ablation than with endovenous laser treatment (EVLT).32 (Author’s note: these studies all utilized ClosurePLUSTM catheters.)
CLOSUREFASTTM — THE FUTURE
The endovenous Closure® procedure has evolved from the original catheter and techniques to the introduction of improved devices (ClosurePLUSTM, ClosureFASTTM) and techniques (tumescence) designed to improve outcomes and reduce the incidence of complications, and newly designed devices (ClosureRFSTM) to broaden applications. The introduction of the ClosureFASTTM catheter in 2007 was intended to substantially shorten procedure time and it appears that it has done just that. The device has a 7 cm heating element that remains stationary during a 20-second energy delivery period. The catheter is then repeatedly retracted 6.5 cm and energized for 20 seconds at each segment until the desired length of vein has been treated. The closureFASTTM catheter has undergone successful early clinical studies. Proebstle et al reported the first multicenter study of 252 GSVs treated in 194 patients. The average energy delivery time was 2.2 minutes over an average 36.7cm vein length; 16.4 minutes average elapsed time from catheter insertion to final removal. Initial vein occlusion was 100%, and life-table analysis out to 6 months indicated a 99.6% occlusion rate.33 In follow-up, Proebstle reported a 96.7% occlusion rate for 223 vein segments at one year.34
The Reno Vein Clinic began using ClosureFASTTM in May 2007, and by November 2007 had treated 138 saphenous vein segments of which 15 were SSV treatments. Within the first six months, only one segment had completely recanalized, and the complication rates were similar to those of the ClosurePLUSTM procedure. However, in several instances there were multiple interruptions of the procedure due to inadequate or uneven heating of the vein segments, which resulted in damage to the heating element portion of six ClosureFASTTM catheters. In November 2007, an adjustment was made in what proved to be a significant improvement to the technique — the limb undergoing treatment was elevated 30-45 degrees from the horizontal to effect complete emptying of the superficial venous system (see Figure 2). DUS demonstrated complete emptying of the saphenous vein with tumescence and elevation. Implementation of this maneuver resolved the uneven heating problem, and the average energy delivered to the vein segments dropped from 110 Joules/cm to 60 Joules/cm (average of two procedure measurements). From November 2007 to October 2008, 131 saphenous vein segments (of which 18 were SSVs) were treated with 100% initial occlusion and absence of hypoesthesia and deep venous thrombosis related to the RF procedure. Interruptions ceased and there were no further episodes of heatrelated catheter damage. One patient suffered a second degree skin burn at the insertion site, probably as a result of inadvertent positioning of the catheter in the introducer sheath during the final segment treatment. Three key points of the ClosureFASTTM technique should be emphasized to assure a smooth and successful treatment of the patient:
1/ To avoid possible thermal injury to the SFJ, the catheter tip must be positioned 2 cm distal to the SFJ, regardless of superficial epigastric vein location. One must reaffirm location by longitudinal and transverse DUS images.
2/ There is a step-up hub located 2 cm behind the heating element; use this indicator to locate the position of the catheter beneath the skin at the insertion site during the final segment treatment. This should help avoid skin burns at this location.
3/ Elevate the limb to be treated, either manually or by table reposition, 30-45 degrees during the short twoto four-minute treatment course. This provides complete emptying of the vein segment and allows a smooth, uninterrupted procedure.
CONCLUSIONS
The evidence published in peer-reviewed journals, four studies of which are level one, suggests that at least out to five years outcomes of RF obliteration of saphenous vein reflux are comparable to those of traditional stripping and ligation. The risks of serious complications such as DVT are low and comparable to those that attend stripping and ligation. Lesser complications, when they do occur, are time limited and usually of minor consequence.
Using the RF Closure® equipment and employing current techniques described in this report, an experienced clinician, modifying details to suit individual clinical settings, can expect the following:
1) 98% to 100% successful initial ablation;
2) less than 1% rate of complications such as common femoral vein clot extension and DVT, temporary sensory thermal nerve injury and second degree thermal skin injury;
and 3) five-year ablation and reflux-free outcomes of >90%. In high-risk patients, eg, the obese, or those on anticoagulation or having comorbidities, the Closure® procedures may be the better treatment option because of the advantages they offer over traditional surgical methods, especially regarding less trauma. In cases where reflux originates distal to the saphenofemoral junction (which can only be appreciated by DUS), the Closure® method is ideally suited.
Neovascularization following this procedure at the saphenofemoral junction appears to occur rarely and may not be a factor in later recurrent varicose veins, a possible distinct advantage in comparison with surgical stripping and high ligation.11,35 The persistent patency found in the superficial epigastric vein and other less frequently seen groin branches and the pattern of failures of the Closure® procedure have been described recently in the five-year report by the Closure® Study Group and the results are encouraging for long-term successful relief from superficial venous hypertension and reflux. There is little reason to doubt that results with the new ClosureFASTTM segmental heating catheter would be any different, if not better; short-term reports support this assertion.34
Radiofrequency obliteration of saphenous vein reflux, with the caveat that it is done by a qualified physician, has become a safe, effective, and preferred alternative to traditional surgical techniques, evidenced by many publications including the Closure® Study Group fiveyear outcomes. Schmedt et al, reporting certain anatomical findings by an innovative research investigational tool, endoluminal optical coherence tomography, demonstrated a more uniform and complete disintegration of the radiofrequency (ClosurePLUSTM, 6Fr)–treated bovine vein compared with endovenous laser–treated bovine vein.14 Whether radiofrequency deserves to be the treatment of choice among endovenous obliteration procedures will require further well-designed randomized studies. In the meantime it makes sense to offer this innovative technology as a primary choice for the patient with saphenous vein reflux of primary origin.
The author wishes to express his gratitude to Jeffrey S. Frisbie for his technical assistance in the preparation of this report.

REFERENCES
2. Filingeri V, Gravante G, Cassisa D. Physics of radiofrequency in proctology. Eur Rev Med Pharmacol Sci. 2005;9:349-354.
3. Brown DB. Concepts, considerations, and concerns on the cutting edge of radiofrequency ablation. J Vasc Interv Radiol. 2005;16:597-613.
4. Gillams AR. The use of radiofrequency in cancer. Br J Cancer. 2005;92:1825-1829.
5. Berjano EJ. Theoretical modeling for radiofrequency ablation: state-of-theart and challenges for the future. Biomed Eng Online. 2006;April 18;5:24.
6. Sadick N, Sorhaindo L. The radiofrequency frontier: a review of radiofrequency and combined radiofrequency pulsed-light technology in aesthetic medicine. Facial Plast Surg. 2005;21:131-138.
7. Chandler JG, Pichot O, Sessa C, et al. Defining the role of extended saphenofemoral junction ligation: A prospective comparative study. J Vasc Surg. 2000;32:941-953.
8. Chandler JG, Pichot O, Sessa C, et al. Treatment of primary venous insufficiency by endovenous saphenous vein obliteration. Vasc Surg. 2000;34:201-214.
9. Nicolini P; Closure Group. Treatment of primary varicose veins by endovenous obliteration with the VNUS closure system: results of a prospective multicentre study. Eur J Vasc Endovasc Surg. 2005;29:433-439.
10. Merchant RF, Pichot O, Myers KA. Four-year follow-up on endovascular radiofrequency obliteration of great saphenous reflux. Dermatol Surg. 2005;31:129-134.
11. Pichot O, Kabnick LS, Creton D, et al. Duplex ultrasound scan findings two years after great saphenous vein radiofrequency endovenous obliteration. J Vasc Surg. 2004;39:189-195.
12. Lurie F, Creton D, Eklof B, et al. Prospective randomised study of endovenous radiofrequency obliteration (closure) versus ligation and vein stripping (EVOLVeS): twoyear follow-up. Eur J Vasc Endovasc Surg. 2005;29:67-73.
13. Manfrini S, Gasbarro V, Danielsson G, et al. Endovenous management of saphenous vein reflux. J Vasc Surg. 2000;32:330-342.
14. Schmedt CG, Meissner OA, Hunger K, et al. Evaluation of endovenous radiofrequency ablation and laser therapy with endoluminal optical coherence tomography in an ex vivo model. J Vasc Surg. 2007;45:1047-1058.
15. Zikorus AW, Mirizzi MS. Evaluation of setpoint temperature and pullback speed on vein adventitial temperature during endovenous radiofrequency energy delivery in an in-vitro model. Vasc Endovascular Surg. 2004;38:167- 174.
16. Merchant RF, DePalma RG, Kabnick LS. Endovascular obliteration of saphenous reflux: A multicenter study. J Vasc Surg. 2002;35:1190-1196.
17. Weiss RA, Weiss MA. Controlled radiofrequency endovenous occlusion using a unique radiofrequency catheter under duplex guidance to eliminate saphenous varicose vein reflux: A 2- year follow-up. Dermatol Surg. 2002;28:38-42.
18. Merchant RF, Frisbie JS, Kistner RL. Endovenous radiofrequency obliteration of saphenous vein reflux. In: Pearce WH, Matsumura JS, Yao JS, eds. Trends in Vascular Surgery 2006. Evanston, IL: Greenwood Academic; 2007:429-442.
19. Merchant RF, Pichot O; Closure study group. Long-term outcomes of endovenous radiofrequency obliteration of saphenous reflux as a treatment for superficial venous insufficiency. J Vasc Surg. 2005;42:502- 509.
20. Rautio T, Ohinmaa A, Perälä J, et al. Endovenous obliteration versus conventional stripping operation in the treatment of primary varicose veins: A randomized controlled trial with comparison of costs. J Vasc Surg. 2002;35:958-965.
21. Lurie F, Creton D, Eklof B, et al. Prospective randomized study of endovenous radiofrequency obliteration (closure procedure) versus ligation and stripping in a selected patient population (EVOLVeS Study). J Vasc Surg. 2003;38:207-214.
22. Stötter L, Schaaf I, Bockelbrink A. Comparative outcomes of radiofrequency endoluminal ablation, invagination stripping, and cryostripping in the treatment of great saphenous vein insufficiency. Phlebology. 2006;21:60-64.
23. Hinchliffe RJ, Ubhi J, Beech A, Ellison J, Braithwaite BD. A prospective randomised controlled trial of VNUS closure versus surgery for the treatment of recurrent long saphenous varicose veins. Eur J Vasc Endovasc Surg. 2006;31:212-218.
24. Perala J, Rautio T, Biancari F, et al. Radiofrequency endovenous obliteration versus stripping of the long saphenous vein in the management of primary varicose veins: 3-year outcome of a randomized study. Ann Vasc Surg. 2005;19:669-672.
25. Whiteley MS, Price BA, Scott MJ, Gallagher TM. Radiofrequency ablation of refluxing great saphenous systems, Giacomini veins, and incompetent perforating veins using VNUS Closure and TRLOP technique. Phlebology. 2003;18:52.
26. Peden E, Lumsden A. Radiofrequency ablation of incompetent perforator veins. Perspect Vasc Surg Endovasc Ther. 2007;19:73-77.
27. Gradman WS. Adjunctive proximal vein ligation with endovenous obliteration of great saphenous vein reflux: does it have clinical value? Ann Vasc Surg. 2007;21:155-158.
28. Boros MJ, O’Brien SP, McLaren JT, Collins JT. High ligation of the saphenofemoral junction in endovenous obliteration of varicose veins. Vasc Endovascular Surg. 2008;42:235-238.
29. Pittaluga P, Chastanet S, Guex JJ. Great saphenous vein stripping with preservation of sapheno-femoral confluence: hemodynamic and clinical results. J Vasc Surg. 2008;47:1300-1304.
30. Frasier K, Giangola G, Rosen R, Ginat DT. Endovascular radiofrequency ablation: a novel treatment of venous insufficiency in Klippel-Trenaunay patients. J Vasc Surg. 2008;47:1339- 1345.
31. Vasquez MA, Wang J, Mahathanaruk M, Buczkowski G, Sprehe E, Dosluoglu HH. The utility of the Venous Clinical Severity Score in 682 limbs treated by radiofrequency saphenous vein ablation. J Vasc Surg. 2007;45:1008- 1014.
32. Puggioni A, Kalra M, Carmo M, Mozes G, Gloviczki P. Endovenous laser therapy and radiofrequency ablation of the great saphenous vein: analysis of early efficacy and complications. J Vasc Surg. 2005;42:488-493.
33. Proebstle TM, Vago B, Alm J, Göckeritz O, Lebard C, Pichot O. Treatment of the incompetent great saphenous vein by endovenous radiofrequency powered segmental thermal ablation: first clinical experience. J Vasc Surg. 2008;47:151-156.
34. Proebstle TM, Vago B, Alm J, Göckeritz O, Lebard C, Pichot O. One year follow-up of radiofrequency segmental thermal ablation (RTFA) of great saphenous veins. Presented at American Venous Forum 20th annual meeting, February 20-23, 2008; Charleston, SC.
35. Kianifard B, Holdstock JM, Whiteley MS. Radiofrequency ablation (VNUS closure) does not cause neovascularisation at the groin at one year: results of a case controlled study. Surgeon. 2006;4:71-74.
