Recurrence of venous thromboembolism and its prevention
Paolo PRANDONI
Department of Medical and Surgical Sciences Department
of Medical and Surgical Sciences Thromboembolism Unit
University of Padua, Italy
ABSTRACT
The risk of recurrent venous thromboembolism (VTE) approaches 40% in all patients after 10 years of follow-up. This risk is higher in patients with permanent risk factors for thrombosis, such as active cancer, prolonged immobilization because of disease, and antiphospholipid antibody syndrome; in patients with idiopathic presentation; and in carriers of several thrombophilic abnormalities, including carriers of AT, protein C or S, increased factor VIII, hyperhomocysteinemia, homozygous carriers of factor V Leiden or prothrombin G20210A variant, and carriers of multiple abnormalities. Patients with permanent risk factors for thrombosis should receive indefinite anticoagulation, consisting of subtherapeutic doses of low-molecular-weight heparin (LMWH) in cancer patients, and oral anticoagulants in all other conditions. Patients with idiopathic VTE, including carriers of thrombophilia, should receive 6 to 12 months of anticoagulation. The decision to discontinue anticoagulation after this period, or to go on with conventional or less intense warfarin treatment, should be individually tailored and balanced against the hemorrhagic risk.
INTRODUCTION
Ten years ago we published the results of a prospective cohort study dealing with the long-term follow-up of more than 300 patients after their first episode of deep venous thrombosis (DVT) of the lower extremities, alone or associated with clinically symptomatic pulmonary embolism (PE).1 All of them had received short anticoagulation treatment, ranging from 3 to 6 months. The cumulative incidence of recurrent thromboembolism was approximately 20% after two years, 25% after five years, and 30% after eight years. Overall, this risk was considerably higher than previously thought. Among the investigated risk factors for recurrences, those associated with the highest hazard ratio were idiopathic presentation, malignancy, and thrombophilia. As a consequence of this and other similar observations,2-9 in the last 10 years there has been an increasing tendency to prolong anticoagulation, adjusting it to individual risk profiles.10,11
Ten years later, we published the results of a new prospective cohort study, dealing with the long-term follow-up of more than 1600 patients with a first episode of DVT and/or PE recruited at several centers in Italy.12 We excluded from the evaluation patients with active cancer, as well as all those with an indication for indefinite anticoagulation. The analysis started at the time of coumarin discontinuation. Surprisingly enough, in spite of the exclusion of patients with active cancer, the cumulative incidence of recurrent VTE was even higher than that reported 10 years earlier, approaching 30% after 5 years, 35% after 8 years, and then increasing to 40% after 10 years (Figure 1). As expected, the recurrence risk was twice as high in patients with idiopathic thrombosis as in those with secondary thrombosis. Much work therefore remains to be done in order to improve the long-term prognosis of patients with acute VTE, especially those with idiopathic thromboembolism.
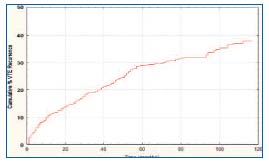
Figure 1. Cummulative incidence of recurrent VTE in patients
with DVT and/or PE.
RISK FACTORS OF RECURRENT THROMBOEMBOLISM
1. Persistent acquired risk factors
Patients with active cancer, especially those with metastatic malignancy and those undergoing chemotherapy, carry a particularly high risk of recurrent VTE after discontinuation of anticoagulation,13 and so do patients with chronic diseases requiring prolonged immobilization.14 Although there is no conclusive evidence from randomized clinical trials, both patient categories should be treated with long-term anticoagulation.
According to the results of recent randomized clinical trials and prospective cohort studies, LMWH in full doses for the first month, followed by a dose ranging from 50% to 100% of the initial regimen, potentially provides a more effective antithrombotic regimen in cancer patients with venous thrombosis than conventional treatment, and is not associated with an increased hemorrhagic risk.15-18 In addition, anticoagulation by LMWH is easier to administer, more convenient and flexible, and not influenced by nutrition problems or liver impairment. Thus, the long-term administration of LMWH should now be considered the treatment of choice in patients with metastatic disease and in those with conditions limiting the use of oral anticoagulants.10,11 Conventional oral anticoagulants are still the treatment of choice in medical conditions other than neoplastic diseases.14
Other conditions associated with a particularly high risk of recurrent VTE and which therefore require indefinite anticoagulation are multiple (especially if idiopathic) VTE episodes,19 the insertion of a permanent vena caval filter (whenever anticoagulation is not contraindicated),20 and antiphospholipid antibody syndrome.10,11 As compared with control subjects, carriers of antiphospholipid antibody syndrome have a higher risk of recurrence,21- 23 and a higher risk of death.23 It is unclear whether20 or not25,26 these subjects require anticoagulation regimens that are more intensive than usual (ie, that produce an international normalized ratio [INR] higher than 3.0). The latest international guidelines advise against the routine adoption of intensive anticoagulation regimens.10,11
2. Idiopathic presentation
Prospective cohort, population-based, and randomized clinical trials of recent years,1-5,8,12,18,24,27-34 have shown that the most important advance in risk assessment of recurrent VTE after cessation of anticoagulant therapy is probably the recognition that patients whose thrombosis is provoked by a major reversible risk factor, such as surgery or major trauma, have a low risk of recurrence, whereas this risk is higher when thrombosis is provoked by a minor reversible risk factor, such as minor leg trauma, estrogen therapy, pregnancy or puerperium, or prolonged air travel, and is particularly high in patients with an idiopathic episode of VTE.
Accordingly, patients with major transient risk factors, such as major trauma or surgery, should be given 12 weeks of anticoagulant therapy.10,11,34 This period can be halved in those patients in whom DVT is confined to the calf vein system.3,29 A longer duration may be considered on a case-by-case basis in patients with minor transient risk factors, such as minor trauma, long air travel, pregnancy, puerperium, or hormonal therapy.14When a thrombotic episode arises during pregnancy, it should be managed with full-dose LMWH for at least three months, bearing in mind that the treatment should not be discontinued before the end of pregnancy, and should always be extended to cover the first six weeks after delivery.10,11 Patients presenting with a first episode of idiopathic VTE should be offered 6 to 12 months of anticoagulation.10,11 Although it has recently been reported that prolonging anticoagulation may offer better protection than confining it to the first 6 months,2 it is generally believed that prolonging anticoagulation is of little value, as it simply delays the date of VTE recurrence while increasing the rate of major bleeding.28-30 The annual incidence of major bleeding during long-term anticoagulation is 1.5-2.0%.35 Moreover, the ‘case-fatality rate’ of an episode of major bleeding is 2 to 3 times as high as it is in patients who develop recurrent VTE.36
Debate surrounds the use of low-dose warfarin to improve the benefit-to-risk ratio of prolonging anticoagulation. In a recent double-blind, randomized trial, Ridker et al convincingly demonstrated that low-intensity warfarin prophylaxis, using a targeted INR of 1.5 to 2.0, is superior to placebo in preventing recurrent venous thromboembolism in patients with idiopathic VTE who have previously been treated for at least three months with warfarin at the conventional dosage, and is not associated with increased hemorrhagic risk.31 However, in a randomized, double-blind trial of similar size, Kearon et al found that low-intensity warfarin (INR, 1.5 to 1.9) was significantly less effective than conventional-intensity warfarin for extended prevention of recurrent thromboembolism in patients with idiopathic VTE, without significant differences in the rate of bleeding complications. Therefore, in patients who require prolonged anticoagulation, a conventional warfarin regimen remains the first-choice treatment.10 However, a low-intensity regimen can be considered in particular situations, depending on individual judgment.11
To optimize the long-term treatment of VTE, new strategies and new drugs are currently under investigation. In a recent prospective cohort study, we have shown that the persistence of residual thrombosis after an episode of proximal DVT, as detected by repeated ultrasonography, is an independent risk factor for recurrent thromboembolism.37 Veins were considered as recanalized in the case of a vein diameter <2.0 mm in a single determination, or <3.0 mm in two consecutive determinations in patients with DVT. Among 313 consecutive patients with proximal DVT, who were followed up prospectively for up to six years after a three- to six-month period of anticoagulation, those with persistent venous obstruction were at a significantly higher risk of recurrence (RR=2.4), after adjustment for thrombophilia and spontaneous clinical presentation. A similar prognostic value of the resolution of the thrombus was observed by Piovella et al in 179 patients with a symptomatic first episode of DVT, and in 104 patients with DVT occurring after hip replacement surgery serially monitored by ultrasonography over a period of 12 months.38 In this regard, the results of a recent prospective study by Young et al are particularly intriguing.39 In follow-up over several years of a cohort of 316 patients with acute DVT, they found a statistically significant higher risk of death in patients with residual thrombus on follow-up ultrasound than in those with earlier vein recanalization (RR=2.8). Although the majority of deaths were due to malignancy, there was a trend towards increased vascular death in the patients with residual thrombus on follow-up ultrasound (RR=4.1). Two recent reviews of clinical trials comparing regimens of anticoagulant therapy, in which ascending phlebography had been used as a tool to assess thrombus evolution, showed an important inverse correlation between thrombus regression and recurrent VTE.40,41 Strategies that include such an assessment of thrombotic burden are intuitively attractive, since a patient can potentially be managed based on the individual course of the thrombotic disease, rather than by broad guidelines alone.
Following the demonstration that a marker of a thrombotic tendency (D-dimer) can be helpful in risk stratification, and thus ultimately in guiding therapy, in individual patients with DVT,7,42-44 Palareti et al performed D-dimer testing one month after the discontinuation of anticoagulation in consecutive patients with a first idiopathic VTE who had received a vitamin K antagonist for at least 3 months.45 Patients with normal D-dimer level did not resume anticoagulation, whereas those with an abnormal D-dimer level were randomly assigned either to resume or to discontinue treatment. Among patients who stopped anticoagulation, the adjusted hazard ratio for recurrent VTE among those with an abnormal Ddimer level, as compared with those with a normal Ddimer level, was 2.3. However, the rate of recurrent VTE in patients with normal D-dimer level who had longterm follow-up was not as low as expected (6.2%). Among the patients with a positive D-dimer level, VTE recurred in 18 of the 120 patients who stopped anticoagulation, as compared with 3 of the 103 who resumed anticoagulation (2.9%; HR=4.3). In conclusion, patients with an abnormal D-dimer level one month after the discontinuation of anticoagulation need to resume anticoagulation, while the optimal course of anticoagulation in patients with a negative D-dimer level remains to be determined.
Finally, new categories of drugs are emerging, which have the potential to simplify the long-term treatment of patients with VTE by obviating the need for periodic laboratory monitoring, while being associated with a favorable benefit-to-risk ratio. They include compounds that inhibit factor Xa, such as idraparinux, rivaroxaban and apixaban, and compounds that inhibit thrombin, such as ximelagatran and dabigatran etexilate. While ximelagtran, a highly effective drug,33 has been withdrawn from the market owing to its potential liver toxicity, the efficacy and safety of the other preparations are currently being assessed by properly controlled investigations.46
In summary, the optimal long-term treatment for patients with idiopathic VTE remains to be determined. The decision to discontinue anticoagulation after the first 6 to 12 months, or to continue with conventional or less intensive warfarin treatment, should be individually tailored and balanced against the hemorrhagic risks. Criteria in support of prolonging anticoagulation are the severity of the initial episode, combined thrombophilic abnormalities (see later), suspected malignant disease, positive family history, residual vein thrombosis on ultrasound tests, lack of contraindications to long-term anticoagulation, and (last but not least) patient preference. Conversely, criteria in support of discontinuing anticoagulation are thrombus location in the calf vein system, relative contraindications to anticoagulation (such as very old age, liver or renal failure, peptic ulcer), earlier vein recanalization, D-dimer negativity and, once again, patient preference. Correctly informed patients should play a pivotal role in making such an important decision concerning their lives.
3. Inherited thrombophilia
It is unclear whether or to what extent carriers of inherited thrombophilia are at higher risk of recurrent VTE. It is generally accepted, although not conclusively demonstrated, that carriers of AT, protein C and S,1,47,48 carriers of hyperhomocysteinemia,49,50 carriers of increased levels of factor VIII or IX,51-53 carriers of multiple abnormalities,8,12 homozygous carriers of factor V Leiden or prothrombin G20210A variant and heterozygous carriers of both mutations54,55 have a recurrence risk that is higher than that of control subjects. Whether heterozygous carriers of factor V Leiden or prothrombin G20210A variant are also at higher risk of recurrence is controversial, as there are data7,12,56-61 and against this association.6,8,24,31,54,55,62-64 Discrepancies among studies may be related to differences in selection of the inception cohort, length of follow-up, initial treatment of the acute thrombotic disorder, duration of treatment, and changes in general management of thrombotic patients.65 As a consequence, it is virtually unknown whether detection of these abnormalities, which are highly prevalent in Western countries, has the potential to identify a subgroup of patients who might benefit from the adoption of individually adjusted prevention strategies following their first thrombotic episode.10,11 Over recent years a good number of prospective cohort and randomized clinical trials have reported data on the long-term outcome of heterozygous carriers of either mutation after discontinuing anticoagulation. We therefore undertook the first systematic review and meta-analysis of available prospective investigations.66 Our meta-analysis indicates that the heterozygous carriage of FVL confers an increased (by about 40%) risk of VTE recurrence, while the risk conferred by the heterozygous carriage of PTM is lower and of uncertain interpretation, as it depends on the method used for its estimation, and ranges between 20% (using the Mantel-Haenszel fixed-effects model) and 36% (using the Der Simonian and Laird random effects model, which takes into greater account the inter-study variability).66
According to the latest international guidelines, carriers of whichever thrombophilia who have a thrombotic episode in conjunction with a promptly detectable transient risk factor for thrombosis should receive shortterm (3 to 6 months) anticoagulation. Heterozygous carriers of factor V Leiden or prothrombin G20210A variant who develop an idiopathic VTE episode should be regarded as noncarriers. Carriers of multiple abnormalities, carriers of AT, protein C or S, increased factor VIII or IX, hyperhomocysteinemia, homozygous carriers of factor V Leiden or prothrombin G20210A variant and heterozygous carriers of both mutations should receive a 6- to 12-month course of anticoagulation.10,11 According to the results of a recent controlled, randomized clinical trial, homocysteine lowering by B-vitamin supplementation does not help prevent recurrent venous thrombosis.67 As patients with thrombophilic defects who belong to thrombophilic families are more likely to have recurrences than are unselected patients with thrombophilic defects,65 this consideration should be taken into account when deciding the duration of anticoagulant therapy following the first thrombotic episode. Further prospective studies addressing the role of thrombophilia in determining the risk of recurrent VTE are indicated, as are randomized studies addressing the benefit-to-risk ratio of prolonging anticoagulation in carriers of thrombophilic abnormalities. Meanwhile, clinical judgment on a case-by-case basis should be used when administering long-term anticoagulation to carriers of whichever thrombophilic abnormality after the first thrombotic episode.
4. Other factors
In a prospective study conducted in Austria, patients with clinically symptomatic PE were found to be associated with a much higher risk of recurrent events than those with symptomatic DVT not associated with PE.27 These findings, however, have not been confirmed by a prospective cohort investigation recently carried out at our institution in a much wider series of patients.12 In our study, the rate of recurrent VTE was significantly higher in patients with proximal DVT (alone or associated with clinically symptomatic PE) than in those with PE alone at presentation (Figure 2).
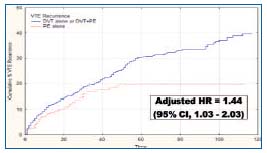
Figure 2. Cumulative incidence of recurrent VTE in patients with primary DVT as compared with those with primary PE.
Accordingly, there seems to be no reason to systematically adopt a longer duration of anticoagulation in patients with PE than in those with DVT. Our results are consistent with data from a randomized, controlled clinical trial, which failed to show an appreciable advantage from prolonging anticoagulation beyond three months both in the subgroup of patients with idiopathic PE and in the subgroup of patients with secondary PE.30 Interestingly, in both the Austrian and Italian cohorts, patients with clinically symptomatic PE were at a higher risk of recurrent PE than those with DVT alone.12,27 As PE is potentially more dangerous than DVT alone, long-term anticoagulation may be considered in selected patients with idiopathic PE, at least in those presenting with life-threatening manifestations.14
Recently, an unexpected association has been reported between male sex and recurrent VTE, especially in patients with idiopathic VTE.68 The are recent literature data for5,6,8,69-73 and against1,2,4,7,74-76 this association. In a prospective study recently conducted in Italy in a broad cohort of patients, we could only find a slight and nonsignificant increase in the risk of recurrent VTE in men.12
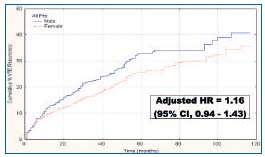
Figure 3. Cumulative incidence of recurrent VTE in males as compared with females.
Even when the analysis was confined to patients with idiopathic VTE, we could not show significant differences between men and women in this regard. Even if we assume that women have a slightly lower recurrence rate than men, the 35% recurrence rate we observed after 10 years of follow-up, as compared with 40% in males, means that this difference is of virtually no clinical relevance. Therefore, based on current evidence, we think that sex should not be regarded as a qualifying factor in deciding the duration of oral anticoagulant therapy after the first episode of thrombosis.
Other predictive factors of VTE recurrence are the location of DVT and the patient’s age. As clearly shown by several investigations, patients with isolated calf DVT have a lower risk of recurrent VTE than patients in whom thrombosis involves the popliteal or more proximal veins.3,29 Accordingly, patients with isolated calf vein thrombosis, except for selected cases, should not receive anticoagulation for longer than 12 weeks when the clinical presentation is idiopathic, or for longer than 6 weeks when the thrombotic episode is triggered by transient risk factors.10,11 Finally, old age, which has long been regarded as a risk factor for venous thrombosis,5 has recently been identified as a predictor of recurrent VTE.12 Thus, the common practice of administering less intensive or shorter term anticoagulation to old patients, because of the fear of hemorrhagic complications, should be reconsidered.77
Women who had their first episode of thromboembolism while of childbearing age are at higher risk of recurrence when they are given hormonal treatment or become pregnant. This risk is particularly high in women in whom the first episode was triggered by hormonal compounds or developed during pregnancy.78-80 Accordingly, hormonal treatment should be strongly discouraged in women with previous VTE. Whenever hormonal treatment is deemed to be necessary, the concomitant administration of oral anticoagulants should be considered. Postpartum anticoagulation is recommended in all women with previous VTE.81 While the systematic use throughout pregnancy of compression hosiery is recommended in all women with previous VTE, antenatal thromboprophylaxis with LMWH should be offered whenever the previous episode was idiopathic or was pregnancy- or estrogen-related, in carriers of thrombophilia, if there is a family history of thrombosis, if there are additional risk factors (such as obesity), and in women with multiple previous VTE episodes.81
Poor quality of vitamin K antagonists may or may not be an additional risk factor for recurrent VTE.82,83 Interestingly, a family history of VTE does not segregate patients into high- or low-risk categories, and is not suitable to identify patients at increased risk of recurrent VTE.84 Obesity, which is a well-known risk factor for venous thrombosis,85,86 does not seem to increase the risk of recurrent VTE.87 Finally, it has recently been reported that a number of simple laboratory tests, such as determination of activated partial thromboplastin time88 and global coagulation assays measuring thrombin generation,89-90 can help identify patients at a lower or higher risk of recurrent VTE. However, these findings need confirmation.
CONCLUSIONS
The risk of recurrent thromboembolism after an episode of VTE is high, and approaches 40% of all patients after 10 years of follow-up. The risk of recurrent VTE is higher in patients with active cancer, prolonged immobilization because of disease, antiphospholipid antibody syndrome, multiple VTE episodes, and the application of a permanent vena caval filter. If there are no contraindications, patients with permanent risk factors for thrombosis should receive indefinite anticoagulation, consisting of subtherapeutic doses of LMWH in patients with cancer, and vitamin K antagonists in all other conditions. The risk of recurrent VTE is definitely higher in patients with idiopathic DVT than in those with secondary DVT. While in the latter group (including carriers of thrombophilic abnormalities) a short duration of anticoagulation (except for pregnant women, requiring full-dose LMWH throughout pregnancy and for the first six weeks after delivery) is all that is needed, patients with idiopathic VTE, including heterozygous carriers of factor V Leiden or prothrombin mutation, should receive 6 to 12 months of anticoagulation. The decision to discontinue anticoagulation after this period, or to continue with conventional or less intensive warfarin treatment should be individually tailored and balanced against the hemorrhagic risks. It remains to be established whether assessing residual vein thrombosis and/or D-dimer values can help define the optimal duration of anticoagulation. Carriers of multiple abnormalities, carriers of AT, protein C or S, increased factor VIII, hyperhomocysteinemia, homozygous carriers of factor V Leiden or prothrombin G20210A variant and heterozygous carriers of both mutations who develop an episode of idiopathic VTE should receive on average a 6- to 12-month course of anticoagulation. Administration of long-term anticoagulation will depend on case-by-case clinical judgment. Patients with proximal DVT are at a higher risk of recurrent VTE than those with isolated calf DVT. Patients with proximal DVT, alone or associated with PE, are at a higher risk of recurrent VTE than those with PE alone. However, patients with clinically symptomatic PE are at higher risk of recurrent PE than those with DVT alone. The risk of recurrent VTE increases with age, and does not substantially differ between men and women. Women of childbearing age are at higher risk of recurrent VTE when they are given hormonal treatment or become pregnant. Hormonal compounds should no longer be administered. While post-partum anticoagulation is recommended in all women with previous VTE, only those in whom the previous episode was idiopathic or was pregnancy- or estrogen-related require antenatal prophylaxis with LMWH, as do carriers of thrombophilia, women with a family history of thrombosis, those with additional risk factors, and those with multiple previous VTE episodes.

REFERENCES
2. Prandoni P, Villalta S, Bagatella P, et al. The clinical course of deep-vein thrombosis. Prospective long-term follow-up of 528 symptomatic patients. Haematologica. 1997;82:423-428.
3. Schulman S, Rhedin AS, Lindmarker P, et al. A comparison of six weeks with six months of oral anticoagulant therapy after a first episode of venous thromboembolism. N Engl J Med. 1995;332:1661-1665.
4. Hansson PO, Sorbo J, Eriksson H. Recurrent venous thromboembolism after deep vein thrombosis. Incidence and risk factors. Arch Intern Med. 2000;1260:769-774.
5. Heit JA, Mohr DN, Silverstein MD, et al. Predictors of recurrence after deep vein thrombosis and pulmonary embolism. A population-based cohort study. Arch Intern Med. 2000;160:761-768.
6. Baglin T, Luddington R, Brown K, Baglin C. Incidence of recurrent venous thromboembolism in relation to clinical and thrombophilic risk factors: prospective cohort study. Lancet. 2003;362:523-526.
7. Palareti G, Legnani C, Cosmi B, et al. Predictive value of D-Dimer test for recurrent venous thromboembolism after anticoagulation withdrawal in subjects with a previous idiopathic event and in carriers of congenital thrombophilia. Circulation. 2003;108:313-318.
8. Christiansen SC, Cannegieter SC, Koster T, et al. Thrombophilia, clinical factors, and recurrent venous thrombotic events. JAMA. 2005;293:2352-2361.
9. Pengo V, Lensing AW, Prins MH, et al. Incidence of chronic thromboembolic pulmonary hypertension after pulmonary embolism. N Engl J Med. 2004;350:2257- 2264.
10. Büller HR, Agnelli G, Hull RD, et al. Antithrombotic therapy for venous thromboembolic disease: the seventh ACCP conference on antithrombotic and thrombolytic therapy. Chest. 2004;126:401S-428S.
11. Nicolaides AN, Fareed J, Kakkar AK, et al. Prevention and treatment of venous thromboembolism. International Consensus Statement. Guidelines according to scientific evidence. Int Angiol. 2006;25:101-161.
12. Prandoni P, Noventa F, Ghirarduzzi A, et al. The risk of recurrent venous thromboembolism after discontinuing anticoagulation in patients with acute proximal deep vein thrombosis or pulmonary embolism. A prospective cohort study in 1626 patients. Haematologica. 2007;91:199-205.
13. Prandoni P, Falanga A, Piccioli A. Cancer and venous thromboembolism. Lancet Oncol. 2005;6:401-410.
14. Kearon C. Long-term management of patients after venous thromboembolism. Circulation. 2004;110(Suppl. 1):10-18.
15. Lee AY, Levine MN, Baker RI, et al. Lowmolecular- weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med. 2003;349:146-153.
16. Meyer G, Marjanovic Z, Valcke J, et al. Comparison of low-molecular-weight heparin and warfarin for the secondary prevention of venous thromboembolism in patients with cancer. Arch Intern Med. 2002;162:1729-1735.
17. Hull RD, Pineo GF, Brant RF, et al. Longterm low-molecular-weight heparin versus usual care in proximal-vein thrombosis patients with cancer. Am J Med. 2006;119:1062-1072.
18. Deitcher SR, Kessler CM, Merli G, et al. Secondary prevention of venous thromboembolic events in patients with active cancer: enoxaparin alone versus initial enoxaparin followed by warfarin for a 180-day period. Clin Appl Thromb Hemost. 2006;12:389-396.
19. Schulman S, Granqvist S, Holmstrom M, et al. The duration of oral anticoagulant therapy after a second episode of venous thromboembolism. N Engl J Med. 1997;336:393-398.
20. Decousus H, Leizorovicz A, Parent F, et al. A clinical trial of vena cava filters in the prevention of pulmonary embolism in patients with proximal deep-vein thrombosis. N Engl J Med. 1998;338:409-415.
21. Khamashta MA, Cuadrado MJ, Mujic F, et al. The management of thrombosis in the antiphospholipid-antibody syndrome. N Engl J Med. 1995;332:993-997.
22. Prandoni P, Simioni P, Girolami A. Antiphospholipid antibodies, recurrent thromboembolism, and intensity of warfarin anticoagulation. Thromb Haemost. 1996;75:859.
23. Schulman S, Svenungsson E, Granqvist S. Anticardiolipin antibodies predict early recurrence of thromboembolism and death among patients with venous thromboembolism following anticoagulant therapy. Am J Med. 1998;104:332-338.
24. Kearon C, Gent M, Hirsh J, et al. A comparison of three months of anticoagulation with extended anticoagulation for a first episode of idiopathic venous thromboembolism. N Engl J Med. 1999;340:901-907.
25. Crowther MA, Ginsberg JS, Julian J, et al. A comparison of two intensities of warfarin for the prevention of recurrent thrombosis in patients with the antiphospholipid antibody syndrome. N Engl J Med. 2003;349:1133-1138.
26. Finazzi G, Marchioli R, Brancaccio V, et al. A randomized clinical trial of highintensity warfarin vs. conventional antithrombotic therapy for the prevention of recurrent thrombosis in patients with the antiphospholipid syndrome. J Thromb Haemost. 2005;3:848-853.
27. Eichinger S, Weltermann A, Minar E, et al. Symptomatic pulmonary embolism and the risk of recurrent venous thromboembolism. Arch Intern Med. 2004;164:92-96.
28. Agnelli G, Prandoni P, Santamaria MG, et al. Three months versus one year of oral anticoagulant therapy for idiopathic deep venous thrombosis. N Engl J Med. 2001;345:165-169.
29. Pinede L, Ninet J, Duhaut P, et al. Comparison of 3 and 6 months of oral anticoagulant therapy after a first episode of proximal deep vein thrombosis or pulmonary embolism and comparison of 6 and 12 weeks of therapy after isolated calf deep vein thrombosis. Circulation. 2001;103:2453-2460.
30. Agnelli G, Prandoni P, Becattini C, et al. Extended oral anticoagulant therapy after a first episode of pulmonary embolism. Ann Intern Med. 2003;139:19-25.
31. Ridker PM, Goldhaber SZ, Danielson E, et al. Long-term, low-intensity warfarin therapy for the prevention of recurrent venous thromboembolism. N Engl J Med. 2003;348:1425-1434.
32. Kearon C, Ginsberg JS, Kovacs MJ, et al. Comparison of low-intensity warfarin therapy with conventional-intensity warfarin therapy for long-term prevention of recurrent venous thromboembolism. N Engl J Med. 2003;349:631-639.
33. Schulman S, Wahlander K, Lundstrom T, et al. Secondary prevention of venous thromboembolism with the oral direct thrombin inhibitor ximelagatran. N Engl J Med. 2003;349:1713-1721.
34. Kearon C, Ginsberg JS, Anderson DR, et al. Comparison of one month with three months of anticoagulation for a first episode of venous thromboembolism associated with a transient risk factor. J Thromb Haemost. 2004;2:743-749.
35. Levine MN, Raskob G, Beyth RJ, et al. Hemorrhagic complications of anticoagulant treatment: the seventh ACCP conference on antithrombotic and thrombolytic therapy. Chest. 2004;126:287S-310S.
36. Douketis JD, Gu CS, Schulman S, et al. The risk of fatal pulmonary embolism after stopping anticoagulant therapy in patients with venous thromboembolism. Ann Intern Med. (in press).
37. Prandoni P, Lensing AWA, Prins MH, et al. Residual venous thrombosis as a predictive factor of recurrent venous thromboembolism. Ann Intern Med. 2002;137:955-960.
38. Piovella F, Crippa L, Barone M, et al. Normalization rates of compression ultrasonography in patients with a first episode of deep vein thrombosis of the lower limbs: association with recurrence and new thrombosis. Haematologica. 2002;87:515-522.
39. Young L, Ockelford P, Milne D, et al. Post treatment residual thrombus increases the risk of recurrent deep vein thrombosis and mortality. J Thromb Haemost. 2006;4:1919- 1924.
40. Gomez-Outes A, Lecumberri R, Lafuente- Guijosa A, et al. Correlation between thrombus regression and recurrent venous thromboembolism. Examining venographic and clinical effects of lowmolecular- weight heparins: a metaanalysis. J Thromb Haemost. 2004;2:1581- 1587.
41. Hull RD, Marder VJ, Mah AF, et al. Quantitative assessment of thrombus burden predicts the outcome of treatment for venous thrombosis: a systematic review. Am J Med. 2005;118:456-464.
42. Palareti G, Legnani C, Cosmi B, et al. Risk of venous thromboembolism recurrence: high negative predictive value of D-dimer performed after oral anticoagulation is stopped. Thromb Haemost. 2002;87:7-12.
43. Eichinger S, Minar E, Bialonczyk C, et al. D-dimer levels and risk of recurrent venous thromboembolism. JAMA. 2003;290:1071-1074.
44. Fattorini A, Crippa L, Viganò D’Angelo S, et al. Risk of deep vein thrombosis recurrence: high negative predictive value of D-dimer performed during oral anticoagulation. Thromb Haemost. 2002;88:162-163.
45. Palareti G, Cosmi B, Legnani C, et al. Ddimer testing to determine the duration of anticoagulation therapy. N Engl J Med. 2006;355:1780-1789.
46. Agnelli G, Becattini C. New anticoagulants. Semin Thromb Hemost. 2006;32:793-802.
47. Van den Belt AG, Sanson BJ, Simioni P, et al. Recurrence of venous thromboembolism in patients with familial thrombophilia. Arch Intern Med. 1997;157:2227-2232.
48. De Stefano V, Simioni P, Rossi E, et al. The risk of recurrent venous thromboembolism in patients with inherited deficiency of natural anticoagulants antithrombin, protein C and protein S. Haematologica. 2006;91:695-698.
49. Den Heijer M, Blom HJ, Gerrits WB, et al. Is hyperhomocysteinaemia a risk factor for recurrent venous thrombosis? Lancet. 1995;345:882-885.
50. Eichinger S, Stümpflen A, Hirschl M, et al. Hyperhomocysteinemia is a risk factor of recurrent venous thromboembolism. Thromb Haemost. 1998;80:566-569.
51. Kyrle PA, Minar E, Hirschl M, et al. High plasma levels of factor VIII and the risk of recurrent venous thromboembolism. N Engl J Med. 2000;343:457-462.
52. Legnani C, Cosmi B, Cini M, et al. High plasma levels of factor VIII and risk of recurrence of venous thromboembolism. Br J Haematol. 2004;124:504-510.
53. Weltermann A, Eichinger S, Bialonczyk C, et al. The risks of recurrent venous thromboembolism among patients with high factor IX levels. J Thromb Haemost. 2003;1:28-32.
54. De Stefano V, Martinelli I, Mannucci PM, et al. The risk of recurrent deep venous thrombosis among heterozygous carriers of both factor V Leiden and the G20210 prothrombin mutation. N Engl J Med. 1999;341:801-806.
55. Margaglione M, D’Andrea G, Colaizzo D, et al. Coexistence of factor V Leiden and factor II A20210 mutations and recurrent venous thromboembolism. Thromb Haemost. 1999;82:1583-1587.
56. Simioni P, Prandoni P, Lensing AWA, et al. The risk of recurrent venous thromboembolism in patients with an Arg506-Gln mutation in the gene for factor V (factor V Leiden). N Engl J Med. 1997;336:399-403.
57. Simioni P, Prandoni P, Lensing AWA, et al. Risk for subsequent venous thromboembolic complications in carriers of the prothrombin or the factor V gene mutation with a first episode of deep-vein thrombosis. Blood. 2000;96:3329-3333.
58. Ridker PM, Miletich P, Stampfer MJ, et al. Factor V Leiden and risks of recurrent idiopathic venous thromboembolism. Circulation. 1995;91:2800-2802.
59. Miles JS, Miletich JP, Goldhaber SZ, et al. G20210A mutation in the prothrombin gene and the risk of recurrent venous thromboembolism. J Am Coll Cardiol. 2001;37:215-218.
60. Santamaria MG, Agnelli G, Taliani MR, et al. Thrombophilic abnormalities and recurrence of venous thromboembolism in patients treated with standardized anticoagulant treatment. Thromb Res. 2005;116:301-306.
61. Ho WK, Hankey G, Quinlan DJ, Eikelboom J. Risk of recurrent venous thrombembolism in patients with common thrombophilia. Arch Intern Med. 2006;166:729-736.
62. Eichinger S, Pabinger I, Stümpflen A, et al. The risk of recurrent venous thromboembolism in patients with and without factor V Leiden. Thromb Haemost. 1997;77:624-628.
63. Eichinger S, Minar E, Hirschl M, et al. The risk of early recurrent venous thromboembolism after oral anticoagulant therapy in patients with the G20210A transition in the prothrombin gene. Thromb Haemost. 1999;81:14-17.
64. Lindmarker P, Schulman S, Sten-Linder M, et al. The risk of recurrent venous thromboembolism in carriers and noncarriers of the G1691A allele in the coagulation factor V gene and the G20210A allele in the prothrombin gene. Thromb Haemost. 1999;81:684-689.
65. Simioni P, Tormene D, Spiezia L, et al. Inherited thrombophilia and venous thromboembolism. Semin Thromb Hemost. 2006;32:700-708.
66. Marchiori A, Mosena L, Prins MH, Prandoni P. The risk of recurrent venous thromboembolism among heterozygous carriers of factor v Leiden or prothrombin G20210A mutation. A systematic review of prospective studies. Haemotologica. 2007;92:1007-1114
67. Den Heijer M, Willems HP, Blom HJ, et al. Homocysteine lowering by B vitamins and the secondary prevention of deep-vein thrombosis and pulmonary embolism. A randomized, placebo-controlled, double blind trial. Blood. 2007;109:139-144.
68. Kyrle PA, Minar E, Bialonczyk C, et al. The risk of recurrent venous thromboembolism in men and women. N Engl J Med. 2004;350:2558-2563.
69. Nieto JA, Monreal M. Recurrent venous thromboembolism in men and women. N Engl J Med. 2004;351:2015-2018.
70. Eriksson H, Lundström T, Wåhlander K, et al. Prognostic factors for recurrence of venous thromboembolism (VTE) or bleeding during long-term secondary prevention of VTE with ximelagatran. Thromb Haemost. 2005;94:522-527.
71. Schulman S, Lindmarker P, Holmstrom M, et al. Post-thrombotic syndrome, recurrence, and death 10 years after the first episode of venous thromboembolism treated with warfarin for 6 weeks or 6 months. J Thromb Haemost. 2006;4:734-742.
72. McRae S, Tran H, Schulman S, Ginsberg J, Kearon C. Effect of patient’s sex on risk of recurrent venous thromboembolism: a meta-analysis. Lancet. 2006;368:371-378.
73. Cushman M, Glynn RJ, Goldhaber SZ, et al. Hormonal factors and risk of recurrent venous thrombosis: the prevention of recurrent venous thromboembolism trial. J Thromb Haemost. 2006;4:2199-203.
74. Murin S, Romano PS, White RH. Related articles, links comparison of outcomes after hospitalization for deep venous thrombosis or pulmonary embolism. Thromb Haemost. 2002;88:407-414.
75. Agnelli G, Becattini C, Prandoni P. Recurrent venous thromboembolism in men and women. N Engl J Med. 2004;351:2015-2018.
76. Gonzalez-Porras JR, Garcia-Sanz R, Alberca I, et al. Risk of recurrent venous thrombosis in patients with G20210A mutation in the prothrombin gene or factor V Leiden mutation. Blood Coagul Fibrinolysis. 2006;17:23-28.
77. Lopez-Jimenez L, Montero M, Gonzalez- Fajardo JA, et al. Venous thromboembolism in very elderly patients: findings from a prospective registry. Haematologica. 2006;91:1046-1051.
78. Hoibraaten E, Qvigstad E, Arnesen H, et al. Increased risk of recurrent venous thromboembolism during hormone replacement therapy—results of the randomized, double-blind, placebocontrolled estrogen in venous thromboembolism trial. Thromb Haemost. 2000;84:961-967.
79. Pabinger I, Grafenhofer H, Kaider A, et al. Risk of pregnancy-associated recurrent venous thromboembolism in women with a history of venous thrombosis. J Thromb Haemost. 2005;3:949-954.
80. De Stefano V, Martinelli I, Rossi E, et al. The risk of recurrent venous thromboembolism in pregnancy and puerperium without antithrombotic prophylaxis. Br J Haematol. 2006;135:386-391.
81. Bates SM, Greer IA, Hirsh J, Ginsberg JS. Use of antithrombotic agents during pregnancy. The seventh ACCP conference on antithrombotic and thrombolytic therapy. Chest. 2004;126:627S–644S.
82. Palareti G, Legnani C, Cosmi B, et al. Poor anticoagulation quality in the first 3 months after unprovoked venous thromboembolism is a risk factor for longterm recurrence. J Thromb Haemost. 2005;3:955-961.
83. Prandoni P, Van Dongen CJJ, Hutten BA, et al. Quality of anticoagulant treatment and risk of subsequent recurrent thromboembolism in patients with deep venous thrombosis. J Thromb Haemost. 2007;5:1555.
84. Hron G, Eichinger S, Weltermann A, et al. Family history for venous thromboembolism and the risk for recurrence. Am J Med. 2006;119:50-53.
85. Stein PD, Beemath A, Olson RE. Obesity as a risk factor in venous thromboembolism. Am J Med. 2005;118:978-980.
86. Glynn RJ, Rosner B. Comparison of risk factors for the competing risks of coronary heart disease, stroke, and venous thromboembolism. Am J Epidemiol. 2005;162:975-982.
87. Romualdi E, Squizzato A, Ageno W. Abdominal obesity and the risk of recurrent deep vein thrombosis. Thromb Res. 2006; e-pub ahead of print.
88. Grand’maison A, Bates SM, Johnston M, et al. “ProC Global”: a functional screening test that predicts recurrent venous thromboembolism. Thromb Haemost. 2005;93:600-604.
89. Hron G, Kollars M, Binder BR, et al. Identification of patients at low risk for recurrent venous thromboembolism by measuring thrombin generation. JAMA. 2006;296:397-402.
90. Hron G, Eichinger S, Weltermann A, et al. Prediction of recurrent venous thromboembolism by the activated partial thromboplastin time. J Thromb Haemost. 2006;4:752-756.
