What is the future of treatment of varices by endovenous procedures?
Bellerive sur Allier, France, 03700
Introduction
Endovenous techniques for varicose vein ablation were introduced clinically 20 years ago. They are now recommended as a first-choice option by the international guidelines.1,2 While the technical evolution seems to have stabilized for thermal techniques, nonthermal techniques are the new trend. What will the new gold-standard treatment be in the years to come?
Classification of endovenous ablation techniques
Garcia et al3 proposed a classification based both on the mode of action (ie, physical or chemical) and on whether there is a necessity for adding tumescent anesthesia. Thus, we can define four types of ablation techniques: (i) thermal ablation with tumescence; (ii) thermal ablation without tumescence; (iii) chemical ablation with tumescence; and (iv) chemical ablation without tumescence.
Thermal ablation techniques with tumescence use heat to ablate the endothelium and delaminate the collagen in the media, which causes some damage to the adventitial layer. Two techniques – radiofrequency ablation and laser ablation with radial fibers using a 1470-nm frequency – are used worldwide, with equivalent immediate- and mid-term results. A third technique is steam ablation, which was introduced more recently and has less clinical validation in studies. Tumescent anesthesia is technically the most demanding and time-consuming part of the surgery.
Each procedure has its drawbacks; for example, with all thermal techniques, burns are a risk because the veins are too close to the skin and need to be deepened by tumescence. In steam ablation, the catheter itself becomes hot and the entry point must be cooled during the procedure. These methods are better adapted to saphenous trunks, are more difficult to use in extra-saphenous varices (except steam, which can easily be used for cross-tortuosity and be applied to superficial varices). On the other hand, long-term studies, such as the 5-year randomized study by Lawaetz et al,4 have proven the safety and efficacy of these methods.
While these techniques are more expensive for the hospital than stripping in terms of immediate costs, they are less expensive than are newer techniques. A recent study performed in Norway showed that steam ablation was the cheapest option.5
Thermal ablation techniques without tumescence can be performed using only local anesthesia at the entry point of the catheter because the procedure itself is not painful. Cryofibrosis, formally known as cryosclerosis, was proposed by Le Pivert and myself in 19816 at a time when the general anesthesia needed for a stripping procedure had a much higher rate of complications than today. We performed a high ligation of the saphenous trunk and its tributaries and then catheterized the vein downward using a cryoprobe with a 3-mm diameter. The tip of the probe was cooled down to -91°C using NO2 gas. We only froze 1 cm of the vein every 3 to 5 cm, which was a mistake because we left segments of the endothelium that were not ablated. Thus, the mid-term results at 18 months showed a 25% to 30% rate of recurrence, which prompted many practitioners to stop using this method. It has been recently used again with improved results. We have performed a feasibility study using a freezing catheter, without high ligation, and the preliminary results were in line with those of tumescent heating techniques (Figure 1). The main advantage of cryofibrosis is the possibility of resterilizing the cryocatheter up to 50 times, which considerably lowers the cost of the procedure for the hospital.
Frullini and Fortuna8 proposed another thermal nontumescent technique, a technique called laser-assisted foam sclerotherapy, which is not purely thermal because it involves heating the medial layer with a Holmium laser fiber, then injecting sclerosing foam into the venous trunk9; however, few studies have been published outside medical meetings. The cost is higher than using a 1470-nm radial fiber. A significant number of patients find the procedure painful, so it has not been widely used.
In these techniques, tumescence is not used to prevent pain or to protect adjacent nerves, but to compress the vein, thus reducing the lumen and decreasing the blood flow. Parsi initiated the use of a long catheter to deliver the foam.10 We added external compression by using an Esmarch bandage in a technique called the alpha technique. The 1- and 3-year follow-up results show that the technique was comparable to first-generation laser and radiofrequency devices.11 This procedure makes sure the vein is empty of blood, thus avoiding the inactivation of the sclerosing agent with blood proteins described by Tessari et al.12 To dispense with the Eshmarch bandage, Cavezzi et al added tumescent compression to the procedure and obtained good rates of medium-term obliteration.13 These techniques are more efficient for small to medium diameter trunks (ie, up to 7 to 8 mm).
These are the most exciting techniques. Mechanochemical ablation is a combination of a mechanical effect to destroy the endothelium with a chemical action using either a liquid or a foam sclerosing agent. Two catheters are available – ClariVein® and Flebogrif®. The ClariVein® device14 is a rotating tip catheter with a built-in battery and electric motor in the handle, and the liquid sclerosing agent is injected when the catheter is retracted. The medium-term follow up studies comparing this method with radiofrequency ablation are available, showing that the occlusion rates are in the same bracket.15 ClariVein® is a single-use device that is more expensive than thermal fibers, whereas the Flebogrif®16 is a passive device where several spikes are expelled from the catheter to ablate the endothelium, while injecting a sclerosing foam. Full ablation of the endothelium is unlikely with this catheter. The long-term occlusion rates may be less favorable with these techniques than the rates achieved with thermal techniques – only time will tell!
Cyanoacrylate glue is the latest technique to be reimbursed in the US. The glue is applied drop by drop through a thin (4 F) catheter, while the vein is compressed with the ultrasound probe. No tumescence and no compression stockings are required. Mid-term results compared favorably with those of radiofrequency ablation in a recently published randomized study.17 Some recurrent inflammatory reactions have been observed and, in rare cases, these reactions have led to redo surgery with surgical ablation of the treated vein. Several brands of glue are commercially available. The cost of the single-use kit is at least three times the cost of radiofrequency or laser catheters.
What is next?
Endovenous techniques are ablative; they aim to close an incompetent venous segment in order to suppress the reflux, which is one etiology of venous hypertension. Thus, we will not expand on potential conservative techniques that may suppress the reflux by repairing the venous valves or replacing them with prosthetic valves. However, when technically feasible, it is logical to save the saphenous trunk. The CHIVA technique (Conservative Hemodynamic Correction of Venous Insufficiency) and the ASVAL concept (Ambulatory Selective Varicose vein Ablation under Local Anesthesia) are mainly used on early-stage varicose veins with saphenous trunk dilatations less than 7 to 8 mm.
The best candidates are transcutaneous ultrasound techniques. We have been working on such devices since 2005 using specific high-intensity focused ultrasound (HIFU) probes. HIFU probes are already being used clinically for cancer removal, glaucoma treatment, and other applications. Two different approaches – thermal transcutaneous ablation and transcutaneous cavitation – have been tested on animals and/or humans.
Thermal transcutaneous ablation was developed from research performed by the Inserm unit – the Thau lab – in Lyon starting in 200518; this technique was licensed to the French company Theraclion®. The Echopulse® device is a robotized instrument. The treatment is planned on an ultrasound image of the vein to be treated, which must be compressed by the probe. A segment of the volume of a grain of rice is heated to 85°C in 20 seconds. The process is repeated until all of the predefined target has been heated.2,3 The probe incorporates a skin cooling system, but tumescent anesthesia is still necessary in the majority of patients. Obermayer19 reported the first clinical results of this system and confirmed its safety and short-term efficiency for closing short segments, such as perforators and recurrences. With the existing device, it is not possible to ablate a saphenous trunk over its entire length, as it would take a very long time and the patient must stay motionless. Future improvements will open more possibilities.
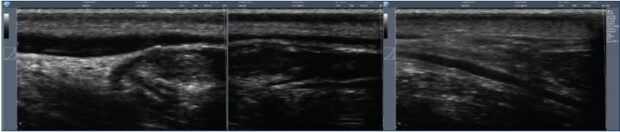
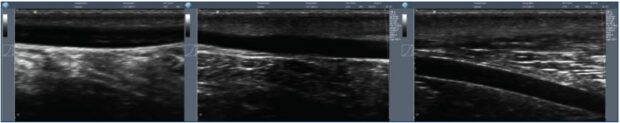
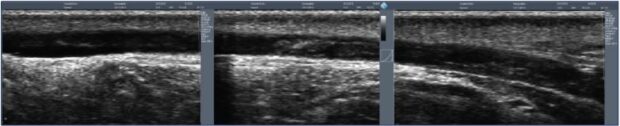
Figure 2. Thermal ultrasound of a sheep saphenous vein before and after ultrasound heating (Veinsound®).
Transcutaneous cavitation devices are being developed by the same Inserm research unit and the Veinsound_ team. Cavitation is a nonthermal biological effect of an ultrasound. Pulses of HIFU cause rapid changes in tissue pressure, which, in liquid blood, generates microbubbles. These cavitation bubbles oscillate and exert shear stress on the surrounding tissue, ie, venous endothelium, and expand rapidly then collapse. Before collapsing, the cavitation bubbles reflect the ultrasound waves, generating more bubbles in a “cavitation cloud.” The energy released ablates the endothelium and damages the media, with a sclerotherapy-like effect being achieved.4,5 The treatment is performed under ultrasound imaging, as it is faster than using thermal ultrasound and painless; tumescence is not necessary.
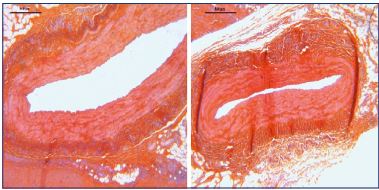
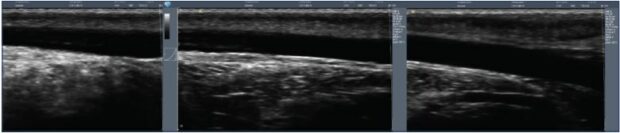
The full length of the saphenous trunks can be treated, as well as superficial varices because there is a lower risk of skin damage. This technique has been tested on sheep (Figures 2-4) and clinical studies are expected to begin before 2020.
Discussion
How will we choose a technique in the near future? Ultrasound techniques are noninvasive (cavitation, thermal ablation without tumescence) or minimally invasive (thermal ablation with tumescence). Thus, an operating theater or dedicated room is no longer necessary, neither is a specialized nurse to help the surgeon. Therefore, health care providers can achieve significant savings.
The economic models for the new techniques are different due to the cost of the equipment. Theraclion® rents out the Echoplulse® HIFU device and the hospital pays a user fee for each treatment performed, covering the rental cost and the single-use accessories (cooling system). This amount should be close to or higher than the cost of a sterile cyanoacrylate glue kit. The Veinsound® cavitation machine is bought by the health care provider, at the price of a high-end echography device. The cost of single-use items is the same as a radial laser fiber kit. Thus, if the hospital performs a high number of treatments, cavitation will lead to more savings compared with endovenous techniques. However, it can be expected that a large number of patients with varicose veins will be treated in-office, leading to a fundamental shift in the medical practice whereby the angiologist will offer a full expertise, from diagnosis to treatment. The time scale of such changes can be fixed around 8 to 10 years.
Ultrasound techniques are at the early stage of clinical application, so randomized comparative studies against endovenous techniques will not be available before 3 to 5 years. In countries where it is possible, reimbursement by state or private insurances will not be considered before mid- to long-term randomized controlled trials are performed, showing noninferiority against the current gold-standard treatment, which is segmental radiofrequency.
Public and private hospitals do not have the same incentives to turn to new techniques. In the state-owned hospitals, more than 80% of patients are still treated with ligation and stripping because surgeons are only allowed this treatment modality for financial reasons. In less developed countries, foam is the treatment of choice. Neither thermal endovenous methods nor the new nontumescent catheters can be competitive on a cost-of-device basis; they will be progressively replaced by transcutaneous methods. Cryofibrosis, using a reusable probe, would be the only option, as it is a less expensive endovenous method and can easily be performed under purely local anesthesia and in a day-surgery organization. In private clinics, who mainly perform laser and radiofrequency ablation, these methods will continue to be the main option until ultrasound techniques are proven to have long-term efficacy in randomized studies. Enrollment in these studies will be difficult because most patients will favor the less aggressive option. Steam and laser-assisted foam sclerotherapy are not widely used, as the companies that introduced these interesting methods are not big enough to finance the studies that would be necessary to convince vascular surgeons and health care providers. This situation may change if they are acquired by larger companies, but it may be too late, as less invasive techniques will come to the market.
Nevertheless, steam ablation will remain a technique of choice in some clinical situations, such as popliteal perforators, where its ability to treat cross-tortuosities is a definite advantage.20 Mechanochemical ablation and glue catheters will remain more expensive for the patient, as they will have to pay for the consumables. The US is a specific case, as glue is now coded and reimbursed by some providers. It may still be used in this country, except if too many explantations must be performed in the years to come. The learning curve is another factor that may help or hinder the adoption of a new technology. Ultrasound therapy is easy to use for specialists who routinely perform echography examinations. Radiologists could then play a significant part and add this treatment modality to their practice.
Conclusion
Endovenous techniques for vein ablation have been used clinically for more than 10 years before becoming the first-choice option to be advocated by the international guidelines. New transcutaneous ultrasound methods will replace them in an unstoppable quest for less aggressive treatments. However, it will take the same amount of time, as it is reasonable to ask for a 5-year follow-up to estimate their real potential.

REFERENCES
1. Gloviczki P, Comerota AJ, Dalsing MC, et al; Society for Vascular Surgery; American Venous Forum. The care of patients with varicose veins and associated chronic venous diseases: clinical practice guidelines of the Society for Vascular Surgery and the American Venous Forum. J Vasc Surg. 2011;53(suppl 5):2S-48S.
2. National Clinical Guideline Centre. Varicose Veins in the Leg: the Diagnosis and Management of Varicose Veins. 2013:1-250.
3. Garcia R, Labropoulos N, Gasparis AP, Elias S. Present and future options for treatment of infrainguinal deep vein disease. J Vasc Surg Venous Lymphat Disord. 2018;6(5):664-671.
4. Lawaetz M, Serup J, Lawaetz B, Bjoern L, Blemings A, Eklof B, Rasmussen L. Comparison of endovenous ablation techniques, foam sclerotherapy and surgical stripping for great saphenous varicose veins. Extended 5-year followup of a RCT. Int Angiol. 2017;36(3):281- 288.
5. Inderhaug E, Schelp CH, Glambek I, Kristiansen IS. Cost-effectiveness analysis of five procedures for great saphenous vein reflux in a Norwegian healthcare setting or societal setting. SAGE Open Med. 2018;6:2050312118801709.
6. Milleret R, Le Pivert P. Cryosclerosis of the saphenous veins in varicose reflux in the obese and elderly. Phlebologie. 1981;34:601-605.
7. Kim KY, Kim JW. Early experience of transilluminated cryosurgery for varicose vein with saphenofemoral reflux: review of 84 patients (131 limbs). Ann Surg Treat Res. 2017;93(2):98-102.
8. Frullini A, Fortuna D. Laser assisted foam sclerotherapy (LAFOS): a new approach to the treatment of incompetent saphenous veins. Phlebologie. 2013;66(1):51-54.
9. Gianesini S, Gafà R, Occhionorelli S, et al. Histologic and sonographic features of holmium laser in the treatment of chronic venous disease. Int Angiol. 2017;36(2):122-128.
10. Parsi K. Catheter-directed sclerotherapy. Phlebology. 2009;24(3):98-107.
11. Milleret R, Garandeau C. Sclérose des grandes veines saphènes à la mousse délivrée par cathéter écho-guidé sur veine vide: alpha-technique bilan des 1.000 premiers traitements. Phlebologie. 2006;59(1):53-56.
12. Tessari L, Izzo M, Cavezzi A, et al. Timing and modality of the sclerosing agents binding to the human proteins: laboratory analysis and clinical evidences. Vein Lymphatics. 2014;3(1):3275.
13. Cavezzi A, Mosti G, Di Paolo S, et al. Ultrasound-guided perisaphenous tumescence infiltration improves the outcomes of long catheter foam sclerotherapy combined with phlebectomy of the varicose tributaries. Vein Lymphatics. 2015;4(1):18-23.
14. Tal MG, Dos Santos SJ, Marano JP, Whiteley MS. Histologic findings after mechanochemical ablation in a caprine model with use of ClariVein. J Vasc Surg Venous Lymphat Disord. 2015;3(1):81-85.
15. Elias S, Raines JK. Mechanochemical tumescentless endovenous ablation: final results of the initial clinical trial. Phlebology. 2012;27(2):67-72.
16. Zubilewicz T, Terlecki P, Terlecki K, Przywara S, Rybak J, Ilzecki M. Application of endovenous mechanochemical ablation (MOCA) with Flebogrif™ to treat varicose veins of the lower extremities: a single center experience over 3 months of observation. Acta Angiologica. 2016;22(4);137-142.
17. Morrison N, Gibson K, McEnroe S, Goldman M, King T, Weiss R, Cher D, Jones A. Randomized trial comparing cyanoacrylate embolization and radiofrequency ablation for incompetent great saphenous veins (VeClose). J Vasc Surg. 2015;61(4):985-994.
18. Pichardo S, Milleret R, Curiel L, Pichot O, Chapelon JY. In vitro experimental study on the treatment of superficial venous insufficiency with high-intensity focused ultrasound. Ultrasound Med Biol. 2006;32(6):883-891.
19. Obermayer A. Ultrasound-guided high-intensity focused ultrasound extracorporeal treatment of superficial lower limb veins: preliminary results and method description. J Vasc Surg Venous Lymphat Disord. 2018;6(4)556-557.
20. Milleret R, Molski M. Steam ablation of Popliteal Perforators. Presented at: 2018 Royal Society of Medicine Venous Forum.